Measure Information Form
Version 2020A2
Measure Information Form
For patients with a stroke due to a cardioembolic source (e.g., atrial fibrillation, mechanical heart valve), warfarin is recommended unless contraindicated. In recent years, novel oral anticoagulants (NOACs) have been developed and approved by the U.S. Food and Drug Administration (FDA) for stroke prevention, and may be considered as an alternative to warfarin for select patients. Anticoagulation therapy is not generally recommended for secondary stroke prevention in patients presumed to have a non-cardioembolic stroke.
Anticoagulants at doses to prevent venous thromboembolism are insufficient antithrombotic therapy to prevent recurrent ischemic stroke or TIA.
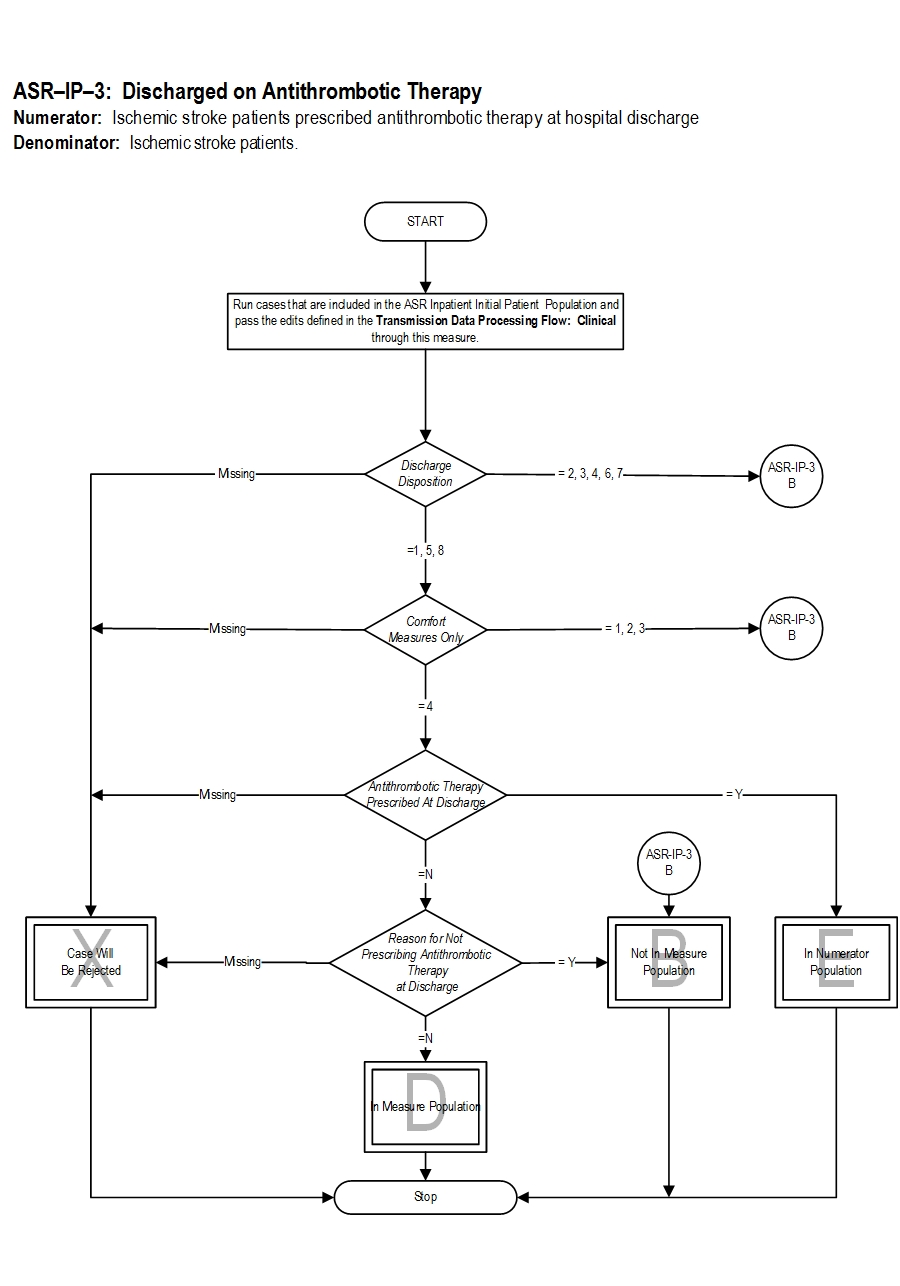
Type Of Measure: Process Improvement Noted As: Increase in the rateIncluded Populations: Not applicable Excluded Populations: None Data Elements:Denominator Statement: Ischemic stroke patients.
Included Populations: Discharges with an ICD-10-CM Principal Diagnosis Code for ischemic stroke as defined in Appendix A, Table 8.1. Excluded Populations:Data Elements:
- Patients less than 18 years of age
- Patients who have a Length of Stay greater than 120 days
- Patients with Comfort Measures Only documented
- Patients discharged to another hospital
- Patients who left against medical advice
- Patients who expired
- Patients discharged to home for hospice care
- Patients discharged to a health care facility for hospice care
- Patients with a documented Reason For Not Prescribing Antithrombotic Therapy at Discharge
- Adams, H., R. Adams, G. Del Zoppo, L. B. Goldstein, Association Stroke Council of the American Heart, and Association American Stroke. "Guidelines for the Early Management of Patients with Ischemic Stroke: 2005 Guidelines Update a Scientific Statement from the Stroke Council of the American Heart Association/American Stroke Association." [In eng]. Stroke 36, no. 4 (Apr 2005): 916-23.
- Adams, H. P., Jr., G. del Zoppo, M. J. Alberts, D. L. Bhatt, L. Brass, A. Furlan, R. L. Grubb, et al. "Guidelines for the Early Management of Adults with Ischemic Stroke: A Guideline from the American Heart Association/American Stroke Association Stroke Council, Clinical Cardiology Council, Cardiovascular Radiology and Intervention Council, and the Atherosclerotic Peripheral Vascular Disease and Quality of Care Outcomes in Research Interdisciplinary Working Groups: The American Academy of Neurology Affirms the Value of This Guideline as an Educational Tool for Neurologists." [In eng]. Stroke 38, no. 5 (May 2007): 1655-711.
- Albers, G. W, P Amarenco, J. D. Easton, R. L. Sacco, and P. Teal. "Antithrombotic and Thrombolytic Therapy for Ischemic Stroke." Chest 119 (2001): 300-20.
- Albers, G. W., P. Amarenco, J. D. Easton, R. L. Sacco, and P. Teal. "Antithrombotic and Thrombolytic Therapy for Ischemic Stroke: The Seventh Accp Conference on Antithrombotic and Thrombolytic Therapy." [In eng]. Chest 126, no. 3 Suppl (Sep 2004): 483S-512S.
- Antithrombotic Trialists, Collaboration. "Collaborative Meta-Analysis of Randomised Trials of Antiplatelet Therapy for Prevention of Death, Myocardial Infarction, and Stroke in High Risk Patients." [In eng]. BMJ 324, no. 7329 (Jan 12 2002): 71-86.
- Bhatt, D. L., K. A. Fox, W. Hacke, P. B. Berger, H. R. Black, W. E. Boden, P. Cacoub, et al. "Clopidogrel and Aspirin Versus Aspirin Alone for the Prevention of Atherothrombotic Events." [In eng]. N Engl J Med 354, no. 16 (Apr 20 2006): 1706-17.
- Brott, T. G., W. M. Clark, S. C. Fagan, J. C. Grotta, L. N. Hopkins, E. C. Jauch, R. E. Latchaw, and S. Starkman. "Stroke: The First Hours. Guidelines for Acute Treatment." National Stroke Association (NSA) (2000).
- Centers for Disease Control and Prevention. "Prevalence and Most Common Causes of Disability among Adults--United States, 2005." [In eng]. MMWR Morb Mortal Wkly Rep 58, no. 16 (May 1 2009): 421-6.
- Chen, Z. M., P. Sandercock, H. C. Pan, C. Counsell, R. Collins, L. S. Liu, J. X. Xie, C. Warlow, and R. Peto. "Indications for Early Aspirin Use in Acute Ischemic Stroke : A Combined Analysis of 40 000 Randomized Patients from the Chinese Acute Stroke Trial and the International Stroke Trial. On Behalf of the Cast and Ist Collaborative Groups." [In eng]. Stroke 31, no. 6 (Jun 2000): 1240-9.
- "Collaborative Overview of Randomised Trials of Antiplatelet Therapy--I: Prevention of Death, Myocardial Infarction, and Stroke by Prolonged Antiplatelet Therapy in Various Categories of Patients. Antiplatelet Trialists' Collaboration." [In eng]. BMJ 308, no. 6921 (Jan 8 1994): 81-106.
- Committee, Caprie Steering. "A Randomised, Blinded, Trial of Clopidogrel Versus Aspirin in Patients at Risk of Ischaemic Events (Caprie). Caprie Steering Committee." [In eng]. Lancet 348, no. 9038 (Nov 16 1996): 1329-39.
- "A Comparison of Two Doses of Aspirin (30 Mg Vs. 283 Mg a Day) in Patients after a Transient Ischemic Attack or Minor Ischemic Stroke. The Dutch Tia Trial Study Group." [In eng]. N Engl J Med 325, no. 18 (Oct 31 1991): 1261-6.
- Coull, B. M., L. S. Williams, L. B. Goldstein, J. F. Meschia, D. Heitzman, S. Chaturvedi, K. C. Johnston, et al. "Anticoagulants and Antiplatelet Agents in Acute Ischemic Stroke: Report of the Joint Stroke Guideline Development Committee of the American Academy of Neurology and the American Stroke Association (a Division of the American Heart Association)." [In eng]. Stroke 33, no. 7 (Jul 2002): 1934-42.
- Diener, H. C., J. Bogousslavsky, L. M. Brass, C. Cimminiello, L. Csiba, M. Kaste, D. Leys, et al. "Aspirin and Clopidogrel Compared with Clopidogrel Alone after Recent Ischaemic Stroke or Transient Ischaemic Attack in High-Risk Patients (Match): Randomised, Double-Blind, Placebo-Controlled Trial." [In eng]. Lancet 364, no. 9431 (Jul 24-30 2004): 331-7.
- Eccles, M., N. Freemantle, and J. Mason. "North of England Evidence Based Guideline Development Project: Guideline on the Use of Aspirin as Secondary Prophylaxis for Vascular Disease in Primary Care. North of England Aspirin Guideline Development Group." [In eng]. BMJ 316, no. 7140 (Apr 25 1998): 1303-9.
- "The European Stroke Prevention Study (Esps). Principal End-Points. The Esps Group." [In eng]. Lancet 2, no. 8572 (Dec 12 1987): 1351-4.
- Farrell, B., J. Godwin, S. Richards, and C. Warlow. "The United Kingdom Transient Ischaemic Attack (Uk-Tia) Aspirin Trial: Final Results." [In eng]. J Neurol Neurosurg Psychiatry 54, no. 12 (Dec 1991): 1044-54.
- Gaspoz, J. M., P. G. Coxson, P. A. Goldman, L. W. Williams, K. M. Kuntz, M. G. Hunink, and L. Goldman. "Cost Effectiveness of Aspirin, Clopidogrel, or Both for Secondary Prevention of Coronary Heart Disease." [In eng]. N Engl J Med 346, no. 23 (Jun 6 2002): 1800-6.
- Gent, M., J. A. Blakely, J. D. Easton, D. J. Ellis, V. C. Hachinski, J. W. Harbison, E. Panak, et al. "The Canadian American Ticlopidine Study (Cats) in Thromboembolic Stroke." [In eng]. Lancet 1, no. 8649 (Jun 3 1989): 1215-20.
- Gorelick, P. B., D. Richardson, M. Kelly, S. Ruland, E. Hung, Y. Harris, S. Kittner, S. Leurgans, and Investigators African American Antiplatelet Stroke Prevention Study. "Aspirin and Ticlopidine for Prevention of Recurrent Stroke in Black Patients: A Randomized Trial." [In eng]. JAMA 289, no. 22 (Jun 11 2003): 2947-57.
- Group, Esprit Study, P. H. Halkes, J. van Gijn, L. J. Kappelle, P. J. Koudstaal, and A. Algra. "Aspirin Plus Dipyridamole Versus Aspirin Alone after Cerebral Ischaemia of Arterial Origin (Esprit): Randomised Controlled Trial." [In eng]. Lancet 367, no. 9523 (May 20 2006): 1665-73.
- Guyatt, G. H., E. A. Akl, M. Crowther, D. D. Gutterman, H. J. Schuunemann, Therapy American College of Chest Physicians Antithrombotic, and Panel Prevention of Thrombosis. "Executive Summary: Antithrombotic Therapy and Prevention of Thrombosis, 9th Ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines." [In eng]. Chest 141, no. 2 Suppl (Feb 2012): 7S-47S.
- Guyatt, G., H. Schunemann, D. Cook, R. Jaeschke, S. Pauker, H. Bucher, and Physicians American College of Chest. "Grades of Recommendation for Antithrombotic Agents." [In eng]. Chest 119, no. 1 Suppl (Jan 2001): 3S-7S.
- Hass, W. K., J. D. Easton, H. P. Adams, Jr., W. Pryse-Phillips, B. A. Molony, S. Anderson, and B. Kamm. "A Randomized Trial Comparing Ticlopidine Hydrochloride with Aspirin for the Prevention of Stroke in High-Risk Patients. Ticlopidine Aspirin Stroke Study Group." [In eng]. N Engl J Med 321, no. 8 (Aug 24 1989): 501-7.
- "The International Stroke Trial (Ist): A Randomised Trial of Aspirin, Subcutaneous Heparin, Both, or Neither among 19435 Patients with Acute Ischaemic Stroke. International Stroke Trial Collaborative Group." [In eng]. Lancet 349, no. 9065 (May 31 1997): 1569-81.
- Jauch, E. C., J. L. Saver, H. P. Adams, Jr., A. Bruno, J. J. Connors, B. M. Demaerschalk, P. Khatri, et al. "Guidelines for the Early Management of Patients with Acute Ischemic Stroke: A Guideline for Healthcare Professionals from the American Heart Association/American Stroke Association." [In Eng]. Stroke (Jan 31 2013).
- Johnson, E. S., S. F. Lanes, C. E. Wentworth, 3rd, M. H. Satterfield, B. L. Abebe, and L. W. Dicker. "A Metaregression Analysis of the Dose-Response Effect of Aspirin on Stroke." [In eng]. Arch Intern Med 159, no. 11 (Jun 14 1999): 1248-53.
- Kennedy, J., M. D. Hill, K. J. Ryckborst, M. Eliasziw, A. M. Demchuk, A. M. Buchan, and Faster Investigators. "Fast Assessment of Stroke and Transient Ischaemic Attack to Prevent Early Recurrence (Faster): A Randomised Controlled Pilot Trial." [In eng]. Lancet Neurol 6, no. 11 (Nov 2007): 961-9.
- Kernan, W.N., B. Ovbiagele, H. R. Black, D. M. Bravata, M. I. Chimowitz, M. D. Ezekowitz, M. C. Fang, M. Fisher, K. L. Furie, D. V. Heck, S. C. Johnston, S. E. Kasner, S. J. Kittner, P. H. Mitchell, M. W. Rich, D. Richardson, L. H. Schwamm, J. A. Wilson. “Guidelines for the Prevention of Stroke in Patients with Stroke and Transient Ischemic Attack: A Guideline for Healthcare Professionals from the American Heart Association/American Stroke Association.” [in eng.] Stroke 45, no. 7 (May 2014): 2160-223.
- Powers WJ, Rabinstein AA, Ackerson T, Adeoye OM, Bambakidis NC, Becker K, Biller J, et al; on behalf of the American Heart Association Stroke Council. 2018 Guidelines for the Early Management of Patients with Acute Ischemic Stroke: A Guideline for Healthcare Professionals From the American Heart Association/American Stroke Association. Stroke. 2018 Jan;49:e45-e46.
- "A Randomized Trial of Aspirin and Sulfinpyrazone in Threatened Stroke. The Canadian Cooperative Study Group." [In eng]. N Engl J Med 299, no. 2 (Jul 13 1978): 53-9.
- Roger, V. L., A. S. Go, D. M. Lloyd-Jones, E. J. Benjamin, J. D. Berry, W. B. Borden, D. M. Bravata, et al. "Heart Disease and Stroke Statistics--2012 Update: A Report from the American Heart Association." [In eng]. Circulation 125, no. 1 (Jan 3 2012): e2-e220.
- Sacco, R. L., H. C. Diener, S. Yusuf, D. Cotton, S. Ounpuu, W. A. Lawton, Y. Palesch, et al. "Aspirin and Extended-Release Dipyridamole Versus Clopidogrel for Recurrent Stroke." [In eng]. N Engl J Med 359, no. 12 (Sep 18 2008): 1238-51.
- "Swedish Aspirin Low-Dose Trial (Salt) of 75 Mg Aspirin as Secondary Prophylaxis after Cerebrovascular Ischaemic Events. The Salt Collaborative Group." [In eng]. Lancet 338, no. 8779 (Nov 30 1991): 1345-9.
- "United Kingdom Transient Ischaemic Attack (Uk-Tia) Aspirin Trial: Interim Results. Uk-Tia Study Group." [In eng]. Br Med J (Clin Res Ed) 296, no. 6618 (Jan 30 1988): 316-20.
CPT® only copyright 2019 American Medical Association. All rights reserved. CPT® is a registered trademark of the American Medical Association.
You, your employees and agents are authorized to use CPT® only as contained in The Joint Commission performance measures solely for your own personal use in directly participating in healthcare programs administered by The Joint Commission. You acknowledge that the American Medical Association (“AMA”) holds all copyright, trademark and other rights in CPT®.
Any use not authorized herein is prohibited, including by way of illustration and not by way of limitation, making copies of CPT® for resale and/or license, transferring copies of CPT® to any party not bound by this Agreement, creating any modified or derivative work of CPT®, or making any commercial use of CPT®. License to use CPT® for any use not authorized herein must be obtained through the American Medical Association, Intellectual Property Services, AMA Plaza, 330 North Wabash Avenue, Suite 39300, Chicago, Illinois 60611-5885. Applications are available at the American Medical Association Web site, www.ama- assn.org/go/cpt.
U.S. Government Rights This product includes CPT® which is commercial technical data, which was developed exclusively at private expense by the American Medical Association, 330 North Wabash Avenue, Chicago, Illinois 60611. The American Medical Association does not agree to license CPT® to the Federal Government based on the license in FAR 52.227-14 (Data Rights - General) and DFARS 252.227-7015 (Technical Data - Commercial Items) or any other license provision. The American Medical Association reserves all rights to approve any license with any Federal agency.
Disclaimer of Warranties and Liabilities. CPT® is provided “as is” without warranty of any kind, either expressed or implied, including but not limited to the implied warranties of merchantability and fitness for a particular purpose. Fee schedules, relative value units, conversion factors and/or related components are not assigned by the AMA, are not part of CPT®, and the (AMA is not recommending their use. The AMA does not directly or indirectly practice medicine or dispense medical services. The responsibility for the content of this product is with The Joint Commission, and no endorsement by the AMA is intended or implied. The AMA disclaims responsibility for any consequences or liability attributable to or related to any use, non-use, or interpretation of information contained or not contained in this product.
This Agreement will terminate upon notice if you violate its terms. The AMA is a third party beneficiary to this Agreement.
Should the foregoing terms and conditions be acceptable to you, please indicate your agreement and acceptance by clicking below on the button labeled “accept”.