Measure Information Form
Version 2020A2
Measure Information Form
The National Consensus Project for Quality Palliative Care (2013) guidelines recommend that the interdisciplinary team assesses and manages pain in a safe and timely manner to a level acceptable to the patient or surrogate and that symptom assessment, treatment, side effect and treatment outcome information should be recorded in the medical record.
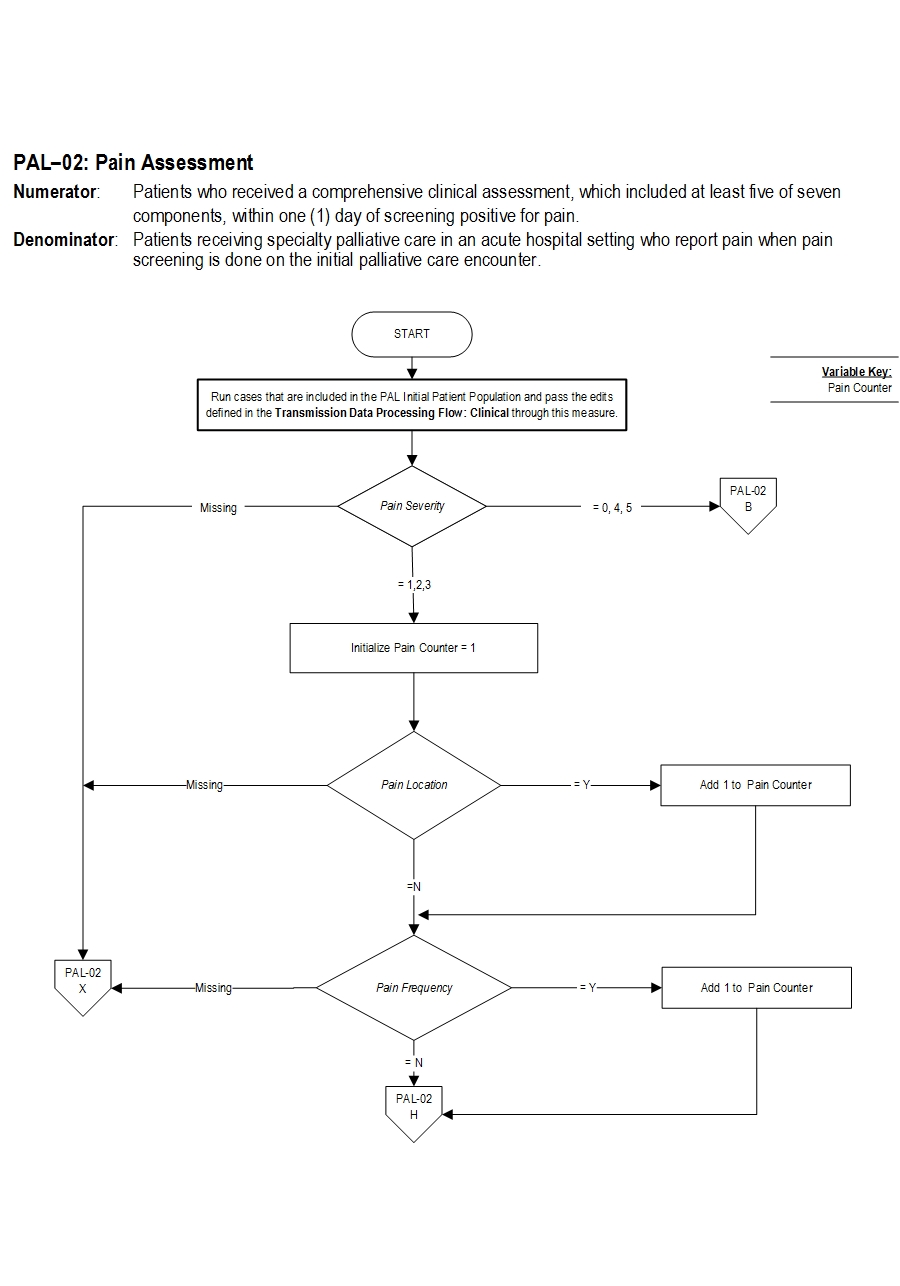
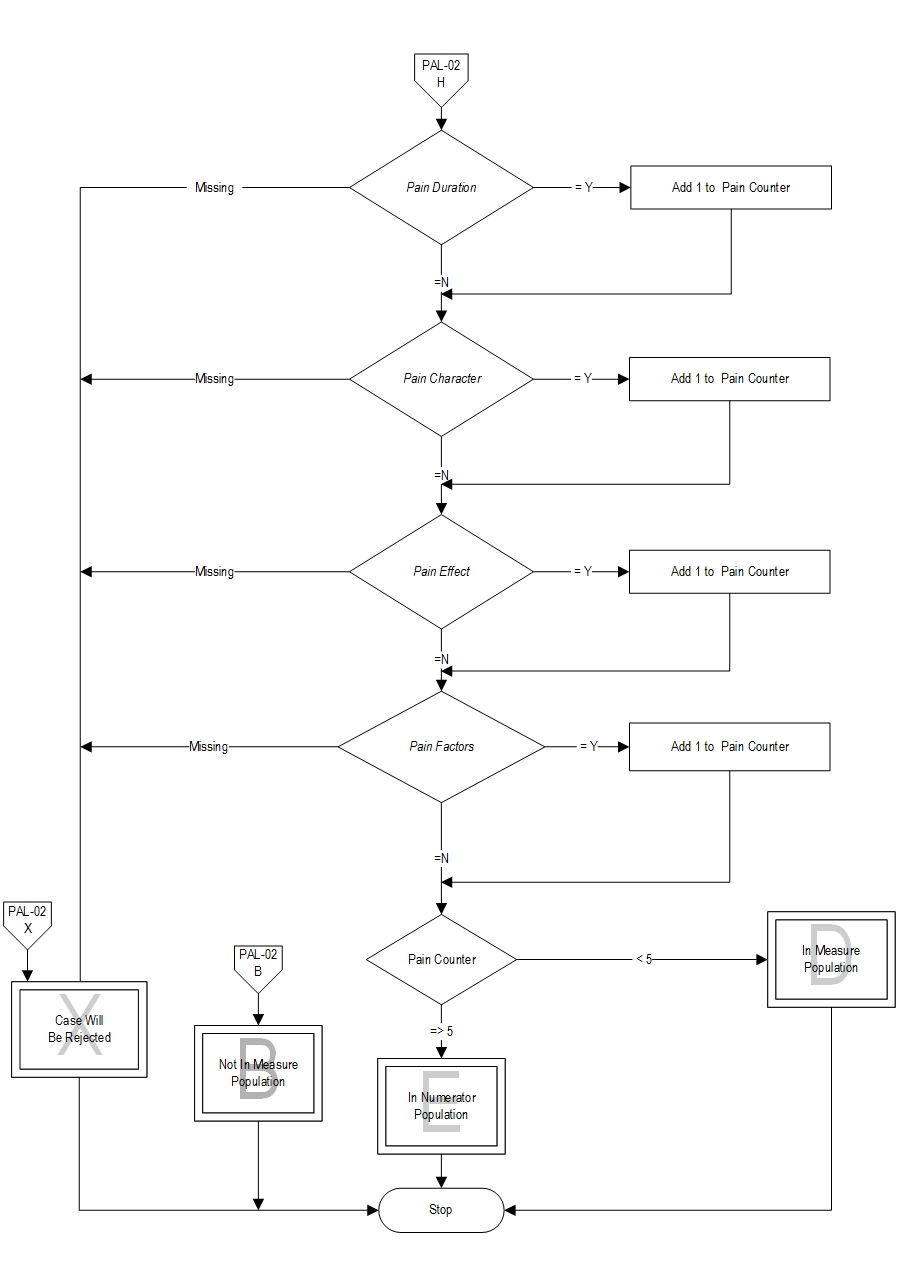
Type Of Measure: Process Improvement Noted As: Increase in the rateIncluded Populations: Not applicable Excluded Populations: None Data Elements:Denominator Statement: Patients receiving specialty palliative care in an acute hospital setting who report pain when pain screening is done on the initial palliative care encounter.
Included Populations: Excluded Populations:Data Elements:
- Palliative care program length of stay less than 1 day
- CMS Hospice Item Set, http://www.cms.gov/Medicare/Quality-Initiatives-Patient-Assessment-Instruments/Hospice-Quality-Reporting/Hospice-Item-Set-HIS.html
- Deandria S, Montanri M, Moja L et al. Prevalence of undertreatment of cancer pain: a review of published literature. Ann Oncol 2008; 19:1985-91.
- Erdek MA, Pronovost PA. Improving assessment and treatment of pain in the critically ill. Int J Qual Health Care 2004; 16:59-64.
- Gordon DB, Dahl JL, Miaskowski C et al. American Pain Society recommendations for improving the quality of acute and cancer pain management. Arch Intern Med 2005; 165:1574-1580.
- Gordon DB, Pelliano TA, Miaskowski C et al. A 10-year review of quality improvement monitoring in pain management: recommendations for standardized outcome measures. Pain Manage Nurs 2002; 4:116-130.
- Measuring What Matters, http://aahpm.org/quality/measuring-what-matters
- Mularski R, White-Chu F, Overbay D et al. Measuring pain as the 5th vital sign does not improve quality of pain management. J Gen Intern Med 2006; 6:607-612.
- National Consensus Project for Quality Palliative Care. Clinical Practice Guidelines for Quality Palliative Care. 3rd ed. Pittsburgh, PA: Author; 2013. http://www.nationalconsensusproject.org/NCP_Clinical_Practice_Guidelines_3rd_Edition.pdf
- PEACE Hospice and Palliative Care Quality Measures, http://www.med.unc.edu/pcare/resources/PEACE-Quality-Measures
- Reynolds K, Henderson M, Schulman A, Hanson LC. Needs of the dying in nursing homes. J Pall Med 2002; 5:895-901.
- Wells N, Pasero C, McCaffery M. Improving the Quality of Care through Pain Assessment and Management. In: Hughes RG, editor. Patient Safety and Quality: An Evidence-Based Handbook for Nurses. Rockville (MD): Agency for Healthcare Research and Quality (US); 2008 Apr. Chapter 17.
American Academy of Hospice and Palliative Care (AAHPM) and Hospice and Palliative Nurses Association (HPNA) Measuring What Matters Project Top Ten Measures That Matter List
CMS Hospice Item Set
PEACE Hospice and Palliative Care Quality Measures Set
CPT® only copyright 2019 American Medical Association. All rights reserved. CPT® is a registered trademark of the American Medical Association.
You, your employees and agents are authorized to use CPT® only as contained in The Joint Commission performance measures solely for your own personal use in directly participating in healthcare programs administered by The Joint Commission. You acknowledge that the American Medical Association (“AMA”) holds all copyright, trademark and other rights in CPT®.
Any use not authorized herein is prohibited, including by way of illustration and not by way of limitation, making copies of CPT® for resale and/or license, transferring copies of CPT® to any party not bound by this Agreement, creating any modified or derivative work of CPT®, or making any commercial use of CPT®. License to use CPT® for any use not authorized herein must be obtained through the American Medical Association, Intellectual Property Services, AMA Plaza, 330 North Wabash Avenue, Suite 39300, Chicago, Illinois 60611-5885. Applications are available at the American Medical Association Web site, www.ama- assn.org/go/cpt.
U.S. Government Rights This product includes CPT® which is commercial technical data, which was developed exclusively at private expense by the American Medical Association, 330 North Wabash Avenue, Chicago, Illinois 60611. The American Medical Association does not agree to license CPT® to the Federal Government based on the license in FAR 52.227-14 (Data Rights - General) and DFARS 252.227-7015 (Technical Data - Commercial Items) or any other license provision. The American Medical Association reserves all rights to approve any license with any Federal agency.
Disclaimer of Warranties and Liabilities. CPT® is provided “as is” without warranty of any kind, either expressed or implied, including but not limited to the implied warranties of merchantability and fitness for a particular purpose. Fee schedules, relative value units, conversion factors and/or related components are not assigned by the AMA, are not part of CPT®, and the (AMA is not recommending their use. The AMA does not directly or indirectly practice medicine or dispense medical services. The responsibility for the content of this product is with The Joint Commission, and no endorsement by the AMA is intended or implied. The AMA disclaims responsibility for any consequences or liability attributable to or related to any use, non-use, or interpretation of information contained or not contained in this product.
This Agreement will terminate upon notice if you violate its terms. The AMA is a third party beneficiary to this Agreement.
Should the foregoing terms and conditions be acceptable to you, please indicate your agreement and acceptance by clicking below on the button labeled “accept”.