Release Notes:
Measure Information Form
Version 2020A2
Measure Set: Comprehensive Stroke (CSTK)
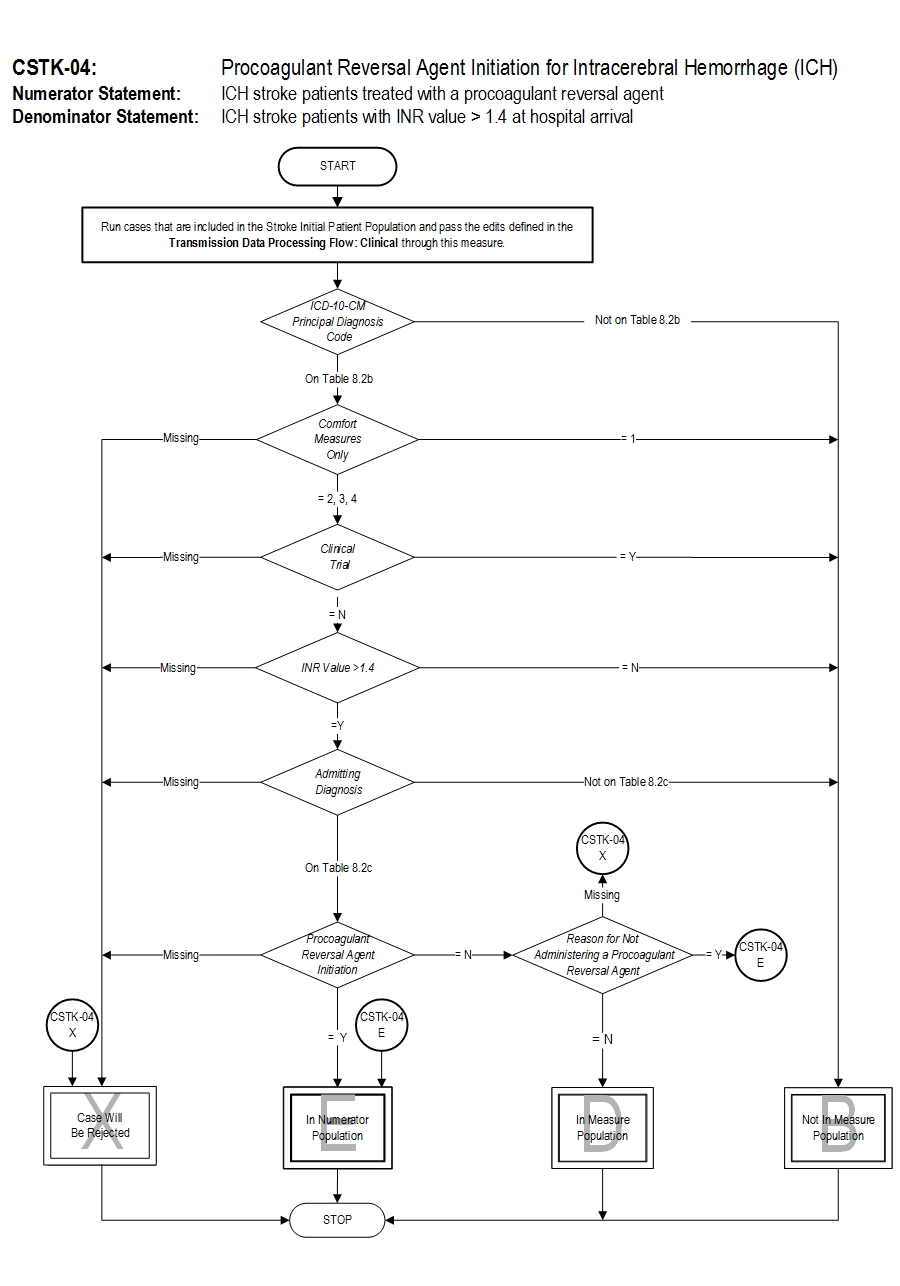
Set Measure ID: CSTK-04
Performance Measure Name: Procoagulant Reversal Agent Initiation for Intracerebral Hemorrhage (ICH )
Description: Intracerebral hemorrhage (ICH) stroke patients with an INR value > 1.4 at hospital arrival who are treated with a procoagulant reversal agent (i.e., fresh frozen plasma, recombinant factor VIIa, prothrombin complex concentrates)
Rationale: Intracerebral hemorrhage (ICH) is a life-threatening disorder. Patients receiving oral anticoagulants (OACs), as well as those with an acquired or congenital coagulopathy, are at increased risk for ICH and hemorrhagic expansion with warfarin-associated bleeds comprising 12% to 15% of all spontaneous hemorrhages. Prompt INR reversal with intravenous infusions of vitamin K and fresh-frozen plasma (FFP) has been historically recommended; however, normalization with prothrombin complex concentrates (PCCs) is increasingly recommended because several studies have shown that these agents can rapidly normalize the INR within minutes. According to the European Union Stroke Initiative (EUSI), patients with oral anticoagulation treatment (OAT) associated ICH and an INR above 1.4, should have OAT discontinued and the INR normalized with PCCs or FFP in addition to intravenous infusion of vitamin K.
Type Of Measure: Process
Improvement Noted As: Increase in the rate
Numerator Statement: ICH stroke patients treated with a procoagulant reversal agent
Included Populations:
As above
Excluded Populations: None
Data Elements:
Denominator Statement: ICH stroke patients with INR value > 1.4 at hospital arrival.
Included Populations:
- Discharges with ICD-10-CM Principal Diagnosis Code for hemorrhagic stroke as defined in Appendix A, Table 8.2b for ICD-10 codes,
AND
- Patients who have an Admitting Diagnosis of primary parenchymal ICH as defined in Appendix A, Table 8.2c for ICD-10 codes,
AND
- INR >1.4 performed closest to hospital arrival
Excluded Populations:
- Patients less than 18 years of age
- Patients who have a Length of Stay > 120 days
- Patients with Comfort Measures Only documented on day of or after hospital arrival
- Patients enrolled in clinical trials
Data Elements:
Risk Adjustment: No.
Data Collection Approach: Retrospective data sources for required data elements include administrative data and medical records.
Data Accuracy: Variation may exist in the assignment of ICD-10 codes; therefore, coding practices may require evaluation to ensure consistency.
Measure Analysis Suggestions: None
Sampling: Yes. Please refer to the measure set specific sampling requirements and for additional information see the Population and Sampling Specifications section.
Data Reported As: Aggregate rate generated from count data reported as a proportion.
Selected References:
1. Ansell J, Hirsch J, Hylek E, Jacobson A, Crowther M, Palareti G; American College of Chest Physicians. Pharmacology and management of the vitamin K antagonists: American College of Chest Physicians Evidence-Based Clinical Practice Guidleines (8th Edition).
Chest. 2008;133(suppl):160S-198S.
2. Fredriksson K, Norrving B, Strömblad, LG. Emergency reversal of anticoagulation after intracerebral hemorrhage.
Stroke. 1992;23:972-977.
3. Frontera JA, Lewin JJ 3rd, Rabinstein AA, Aisiku IP, Alexandrov AW, Cook AM, del Zoppo GJ, Kumar MA, Peerschke EI, Stiefel MF, Teitelbaum JS, Wartenberg KE, Zerfoss CL. Guideline for reversal of antithrombotics in intracranial hemorrhage: a statement for healthcare professionals from the Neurocritical Care Society and Society of Critical Care Medicine.
Neurocrit Care. 2016;24(1):6-46.
4. Goldstein JN, Thomas SH, Frontiero V, Joseph A, Engel C, Snider R, Smith EE, Greenberg SM, Rosand J. Timing of fresh frozen plasma administration and rapid correction of coagulopathy in warfarin-related intracerebral hemorrhage.
Stroke. 2006;37:151-155.
5. Hanley JP. Warfarin reversal.
J Clin Pathol. 2004;57:1132-1139.
6. Hemphill JC III, Greenberg SM, Anderson CS, Becker K, Bendok BR, Cushman M, Fung GL, Goldstein JN, Macdonald L, Mitchell PH, Scott PA, Selim MH, Woo D. Guidelines for the management of spontaneous intracerebral hemorrhage:a guideline for healthcare professionals from the American Heart Association/American Stroke Association.
Stroke. 2015;46:1-29.
7. Leifer D, Bravata DM, Connors JJ III, Hinchey JA, Jauch EC, Johnston SC, Latchaw R, Likosky W, Ogilvy C, Qureshi AI, Summers D, Sung GY, Williams LS, Zorowitz R, on behalf of the American Heart Association Special Writing Group of the Stroke Council, Atherosclerotic Peripheral Vascular Disease Working Group and Council on Cardiovascular Surgery and Anesthesia, and Council on Cardiovascular Nursing. Metrics for measuring quality of care in comprehensive stroke centers: detailed follow-up to Brain Attack Coalition comprehensive stroke center recommendations: a statement for healthcare professionals from the American Heart
Association/American Stroke Association.
Stroke. 2011;42:865-866.
8. Leissinger CA, Blatt PM, Hoots WK, Ewenstein B. Role of prothrombin complex concentrates in reversing warfarin anticoagulation: a review of the literature.
Am J Hematol. 2008;83:137-143.
9. Morgenstern LB, Hemphill JC III, Anderson C, Becker K, Broderick JP, Connolly ES Jr, Greenberg SM, Huang JN, Macdonald RL, Messé SR, Mitchell PH, Selim M, Tamargo RJ; and on behalf of the American Heart Association Stroke council and Council on Cardiovascular Nursing. Guidelines for the management of spontaneous intracerebral hemorrhage: a guideline for healthcare professionals from the American Heart Association/American Stroke Association.
Stroke. 2010;41:2111-2114.
10. Nilsson OG, Lindgren A, Ståhl N, Brandt L, Säveland H. Incidence of intracerebral and subarachnoid hemorrhage in southern Sweden.
J Neurol Neurosurg Psychiatry. 2000;69:601-607.
11. Pabinger I, Brenner B, Kalina U, Knaub S, Nagy A, Ostermann H; Beriplex P/N Reversal Study Group. Prothrombin complex concentrate (Beriplex P/N) for emergency anticoagulation reversal: a prospective multinational clinical trial.
J Thromb Haemost. 2008;6:622-631.
12. Rådberg JA, Olsson JE, Rådberg CT. Prognostic parameters in spontaneous intracerebral hematomas with special reference to anticoagulant treatment.
Stroke. 1991;22:571-576.
13. Reiss H, Meier-Hellman A, Motsch J, Elias M, Kursten FW, Dempfle CE. Prothrombin complex concentrate (Octaplex) in patients requiring immediate reversal of oral anticoagulation.
Thromb Res. 2007;121:9-16.
14. Rosovsky RP, Crowther, MA. What is the evidence for the off-label use of recombinant factor VII (rFVIIa) in the acute reversal of warfarin?
Hematology Am Soc Hematol Educ Program. 2008:36-38.
15. Sjöblom L, Hårdemark HG, Lindgren A, Norrving B, Fahlén M, Samuelsson M, Stigendal L, Stockelberg D, Taghavi A, Wallrup L, Wallvik J. Mangement and prognostic features of intracerebral hemorrhage during anticoagulant therapy: a Swedish multicenter study.
Stroke. 2001;32:2567-2574.
16. Steiner T, Kaste M, Katse M, Forsting M, Mendelow D, Kwiecinski H, Szikora I, Juvela S, Marchel A, Chapot R, Cognard C, Unterberg A. Hacke W. Recommendations for the management of intracranial haemorrhage - part I: spontaneous intracerebral haemorrhage. The European Stroke Initiative Writing Committee and the Writing Committee for the EUSI Executive Committee.
Cerebrovascular Diseases. 2006;22(4):294-316.
17. Watson HG, Baglin T, Laidlaw SL, Makris M, Preston FE. A comparison of the efficacy and rate of response to oral and intravenous vitamin K in reversal of over-anticoagulation with warfarin.
Haematol. 2001;115:145-149.
Measure Algorithm: