Transmission Population Data Processing Flow TJC
Version 2020A1
Transmission Data Processing Flow: Population and Sampling
Introduction
This section contains information regarding the order in which the Joint Commission's Data Warehouse evaluate submitted files, which contain aggregate population and sampling counts. Transmission of population and sampling counts are used to assist in evaluating completeness of submission in accordance with The Joint Commission's sampling requirements.
Each case that is rejected by the process will be listed on a report along with a brief description of the problem. Vendors will access the Joint Commission's HCD Reports via the Performance Measurement System Extranet Track (PET).
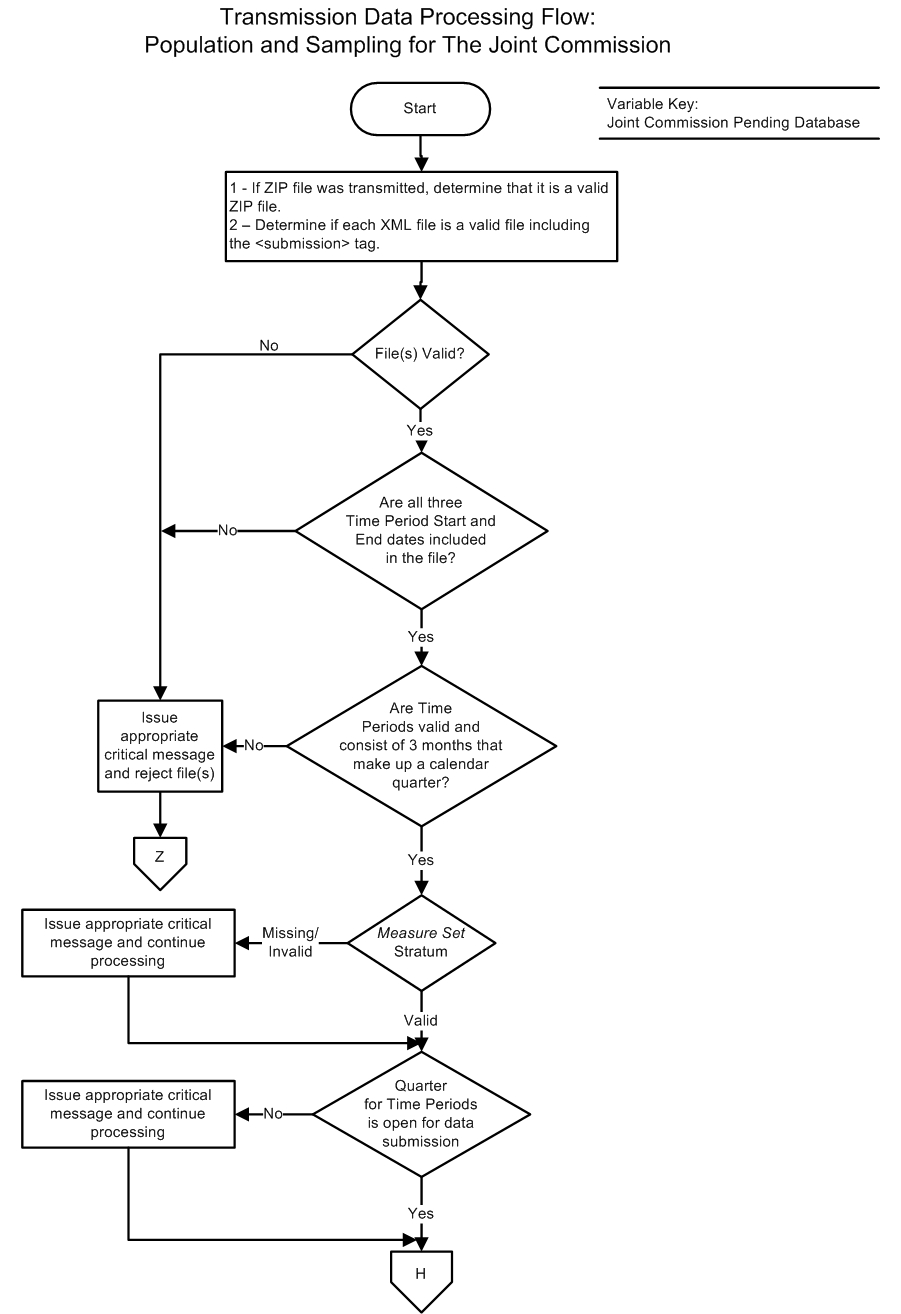
Transmission Data Processing Flow
All data transmitted pass through the following process:- If appropriate, files are verified to be proper zip and XML files.
- If the files are invalid, reject the file(s) and stop processing.
- If the files are valid, continue processing.
Starting with this step, processing is per XML file:
- Data are evaluated to ensure that three individual time periods, which make up a calendar quarter, exist within the file.
- If the data are not expected, reject the XML file and stop processing.
- If the data are expected, continue processing.
- The Measure Set /Stratum is evaluated to ensure a valid value is submitted.
- If the data are not expected, reject the XML file and stop processing.
- If the data are expected, continue processing.
- Data are evaluated to ensure the quarter for Time Periods is open for data submission.
- If the Data Collection quarter is closed, reject the XML file and stop processing.
- If the Data Collection quarter is open, continue processing.
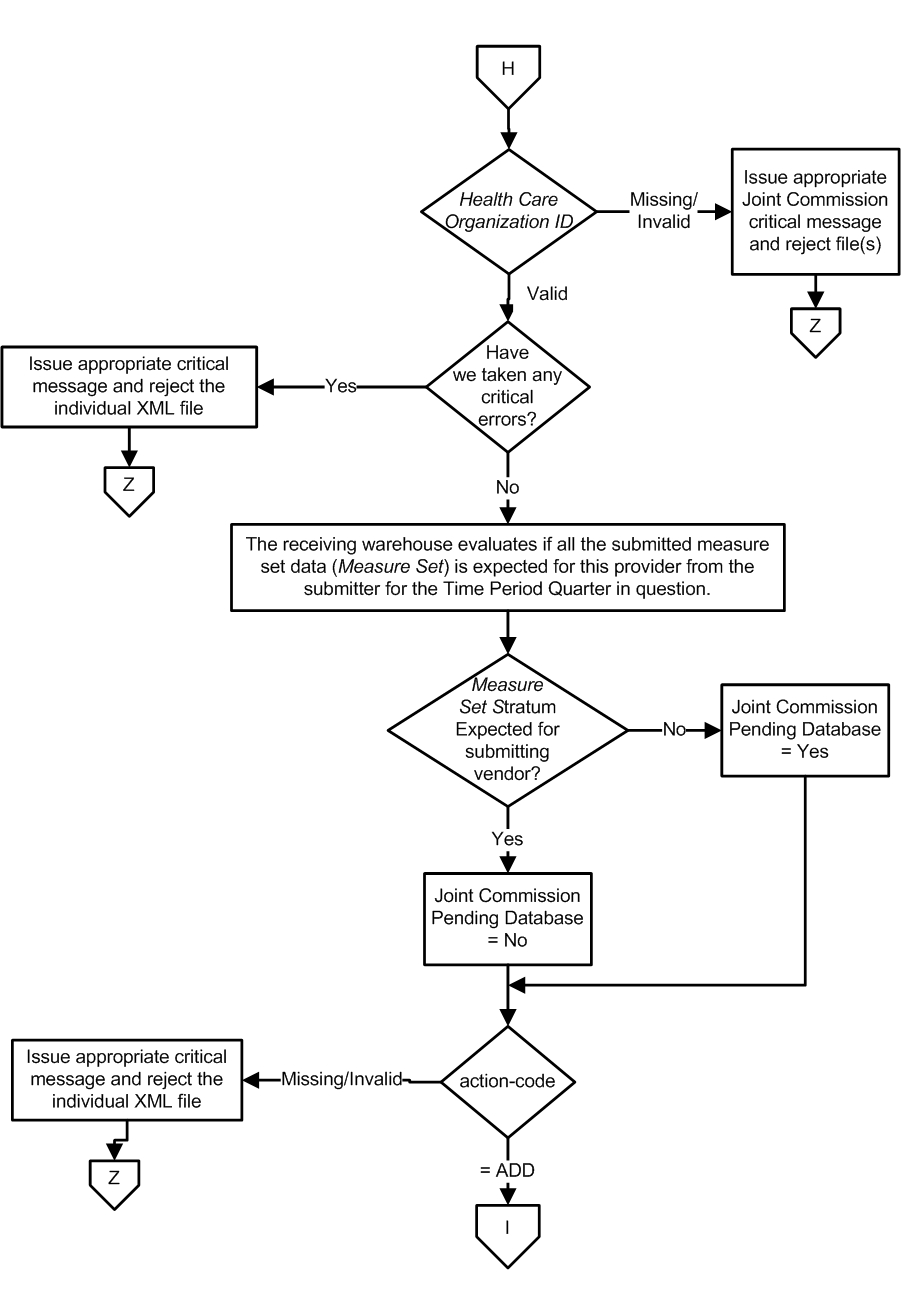
- If the files are submitted to The Joint Commission: The Health Care Organization Identifier is evaluated to ensure a valid value is submitted.
- If the data are not expected, reject the XML file and stop processing.
- If the data are expected, continue processing.
- If the files are submitted to The Joint Commission: Data are evaluated to ensure the Measure Set(s) or Strata are expected from the submitter.
- If the data are not expected, set the reject 'Pending Database' flag = Yes, continue processing.
- If the data are expected, set the reject 'Pending Database' flag = No, continue processing.
- Check the action-code
- If action-code equals Add, continue with processing.
- If the action-code is missing or invalid, reject the XML file and stop processing..
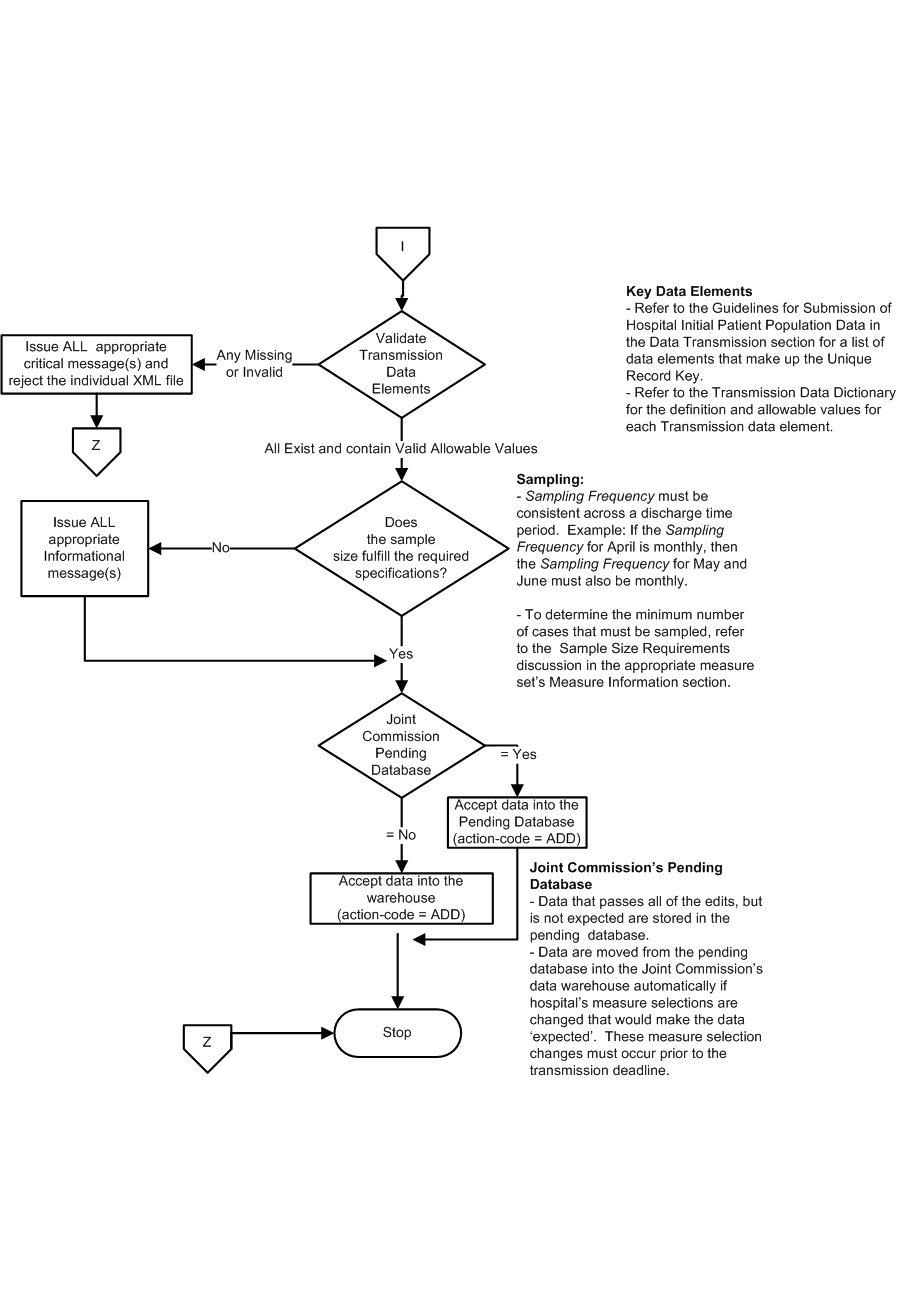
- The transmission data elements, as defined in the Transmission Alphabetical Data Dictionary, are evaluated to ensure they exist and contain valid allowable values. These transmission data elements are required for all submitted files.
- If any transmission data elements fall outside of the data integrity checks, reject the XML file and stop processing.
- If any transmission data element is missing or invalid, reject the XML file and stop processing.
- If all transmission data elements exist and contain valid allowable values, continue processing.
- Data are evaluated to ensure that the sample size fulfills the required specifications. Sample Frequency must be consistent across all three time periods within a calendar quarter.
- If the data are not expected, informational messages generated, continue processing.
- If the data are expected, continue processing.
- a. Check the Pending Database Flag
- If the Pending Database flag = No, the case is accepted into the Joint Commission's Data Warehouse.
- If the Pending Database flag = Yes, the case is accepted into the Joint Commission's Data Warehouse automatically if hospital's measure selections are changed that would make the data 'expected' (refer to step #6b above). These measure selection changes must occur prior to the transmission deadline.
Transmission Data Processing Flow Algorithm
Related Topics
| Related Topics |
|---|
| Table of Contents |
| Transmission Alpha Data Dictionary |
| Transmission Data Processing Flow: Clinical |
| Transmission of Data |
CPT® only copyright 2019 American Medical Association. All rights reserved. CPT® is a registered trademark of the American Medical Association.
You, your employees and agents are authorized to use CPT® only as contained in The Joint Commission performance measures solely for your own personal use in directly participating in healthcare programs administered by The Joint Commission. You acknowledge that the American Medical Association (“AMA”) holds all copyright, trademark and other rights in CPT®.
Any use not authorized herein is prohibited, including by way of illustration and not by way of limitation, making copies of CPT® for resale and/or license, transferring copies of CPT® to any party not bound by this Agreement, creating any modified or derivative work of CPT®, or making any commercial use of CPT®. License to use CPT® for any use not authorized herein must be obtained through the American Medical Association, Intellectual Property Services, AMA Plaza, 330 North Wabash Avenue, Suite 39300, Chicago, Illinois 60611-5885. Applications are available at the American Medical Association Web site, www.ama- assn.org/go/cpt.
U.S. Government Rights This product includes CPT® which is commercial technical data, which was developed exclusively at private expense by the American Medical Association, 330 North Wabash Avenue, Chicago, Illinois 60611. The American Medical Association does not agree to license CPT® to the Federal Government based on the license in FAR 52.227-14 (Data Rights - General) and DFARS 252.227-7015 (Technical Data - Commercial Items) or any other license provision. The American Medical Association reserves all rights to approve any license with any Federal agency.
Disclaimer of Warranties and Liabilities. CPT® is provided “as is” without warranty of any kind, either expressed or implied, including but not limited to the implied warranties of merchantability and fitness for a particular purpose. Fee schedules, relative value units, conversion factors and/or related components are not assigned by the AMA, are not part of CPT®, and the (AMA is not recommending their use. The AMA does not directly or indirectly practice medicine or dispense medical services. The responsibility for the content of this product is with The Joint Commission, and no endorsement by the AMA is intended or implied. The AMA disclaims responsibility for any consequences or liability attributable to or related to any use, non-use, or interpretation of information contained or not contained in this product.
This Agreement will terminate upon notice if you violate its terms. The AMA is a third party beneficiary to this Agreement.
Should the foregoing terms and conditions be acceptable to you, please indicate your agreement and acceptance by clicking below on the button labeled “accept”.