Measure Information Form
Version 2019A1
Measure Information Form
| Set Measure ID | Performance Measure Name |
|---|---|
| THKR-IP-2a | Postoperative Ambulation on Day of Surgery - Hip & Knee Overall |
| THKR-IP-2b | Postoperative Ambulation on Day of Surgery - Hip |
| THKR-IP-2c | Postoperative Ambulation on Day of Surgery - Knee |
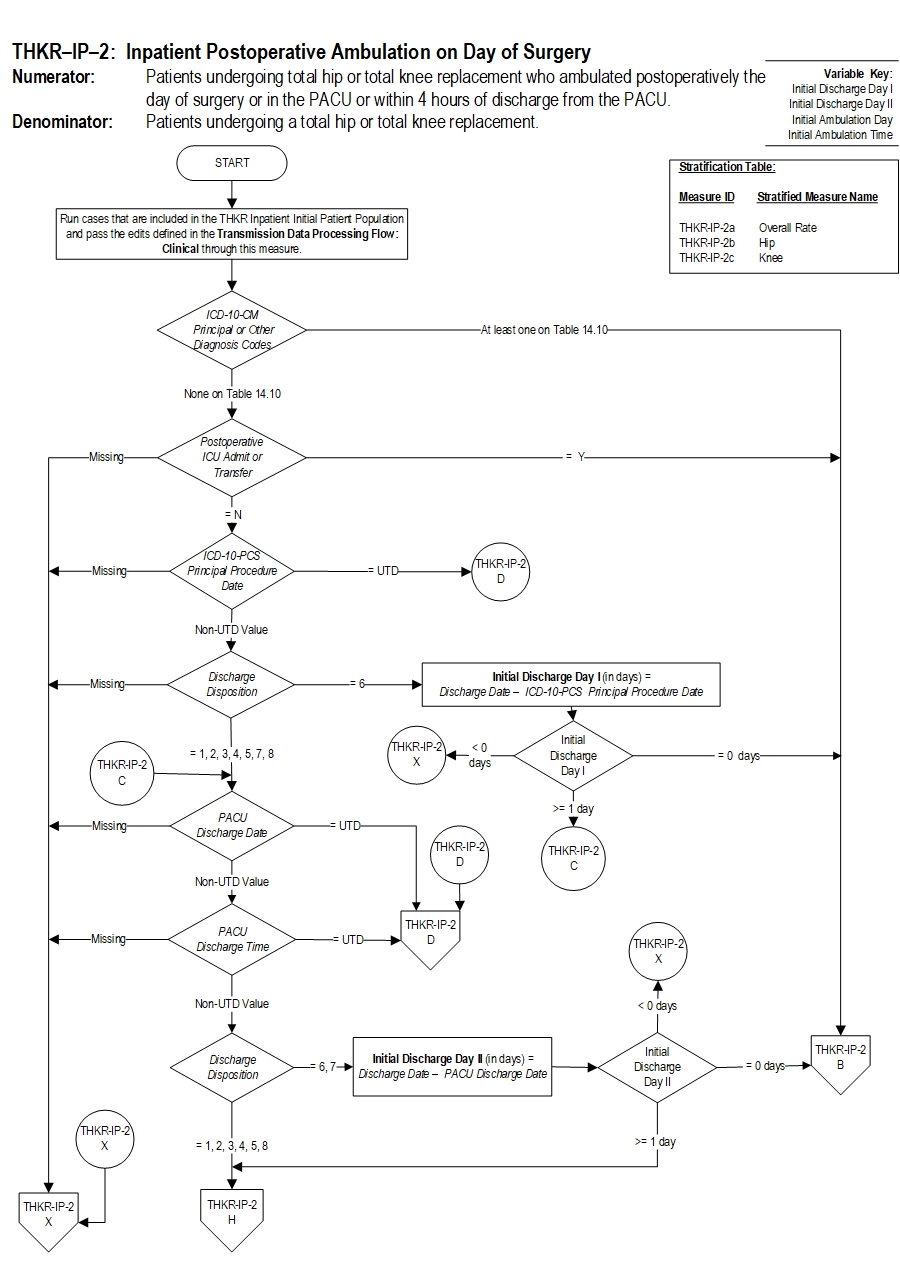
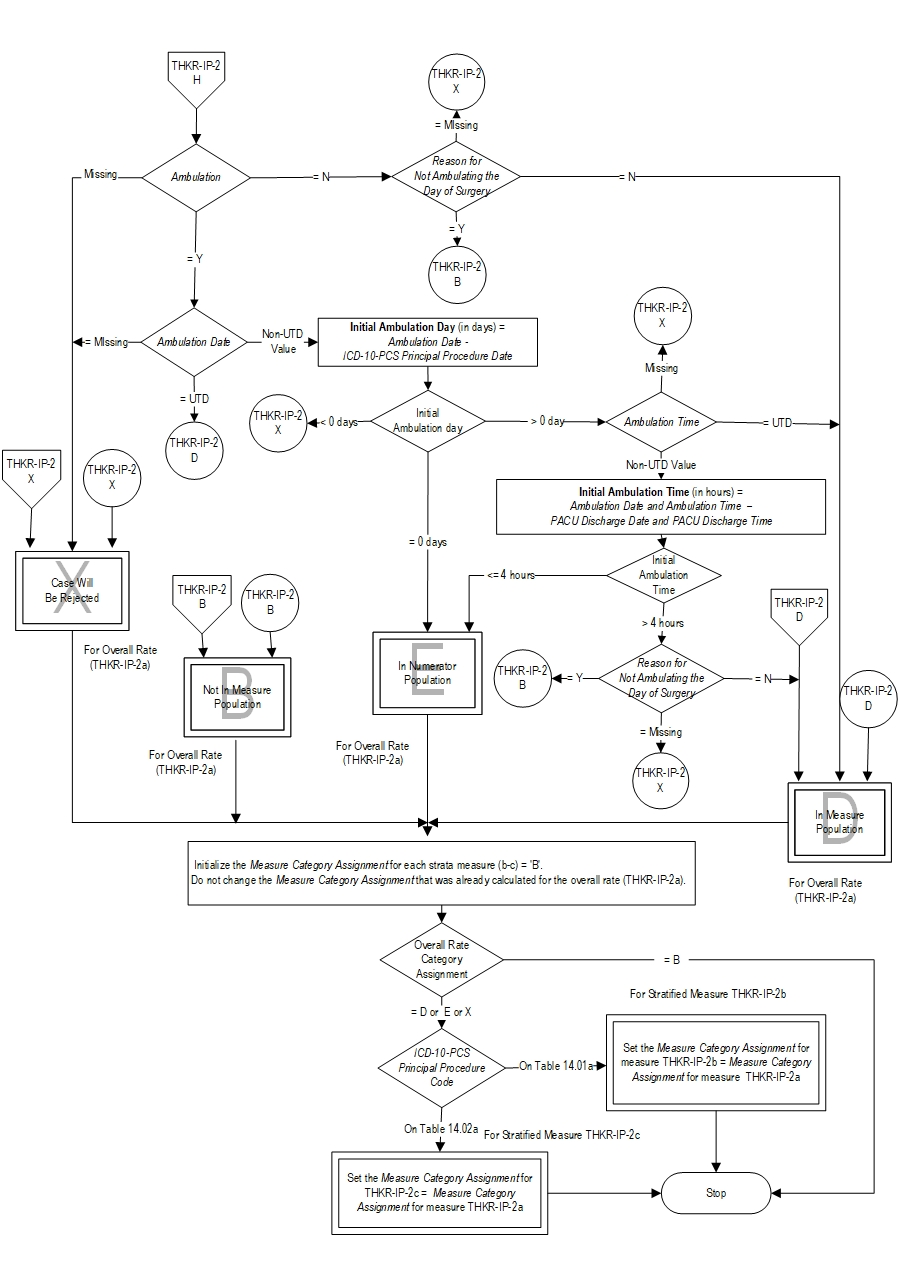
Included Populations:Denominator Statement: Patients undergoing a total hip or total knee replacement.Excluded Populations: None Data Elements:
- Postoperative ambulation (walking) on the day of surgery
- Postoperative ambulation (walking) in the PACU or within 4 hours of discharge from the PACU
Included Populations:Risk Adjustment: No. Data Collection Approach: Retrospective data sources for required data elements include administrative data and medical records. Some hospitals may prefer to gather data concurrently by identifying patients in the population of interest. This approach provides opportunities for improvement at the point of care/service. However, complete documentation includes the principal or other ICD-10 diagnosis and procedure codes, which require retrospective data entry. Data Accuracy: Variation may exist in the assignment of ICD-10 codes; therefore, coding practices may require evaluation to ensure consistency. Measure Analysis Suggestions: None Sampling: Yes. Please refer to the measure set specific sampling requirements and for additional information see the Population and Sampling Specifications Section. Data Reported As: Aggregate rate generated from count data reported as a proportion. Proportion for hip replacements, proportion for knee replacements and aggregated proportion for hip & knee replacements. Selected References:Excluded Populations:
- Patients with an ICD-10-PCS Principal Procedure Code as defined in Appendix A: Table 14.01a Total Hip Replacement or Table 14.02a Total Knee Replacement
Data Elements:
- Patients less than 18 years of age
- Patients who have a Length of Stay greater than 120 days
- Patients with an ICD-10-PCS Other Procedure Code as defined in Appendix A: Table 14.05a (partial hip and partial knee replacements), or Table 14.06a (revision and resurfacing procedures, or Table 14.07a (removal of implanted devices/prostheses)
- Patients with an ICD-10-CM Principal Diagnosis Code or ICD-10-CM Other Diagnosis Code as defined in Appendix A: Table 14.08 (complication of Internal Fixation Device/Prosthesis) or Table 14.09 (malignant neoplasm of the pelvis, sacrum, coccyx, lower limbs, or bone/bone marrow or a disseminated malignant neoplasm), or Table 14.10 (femur, hip, pelvic fracture)
- Postoperative patients who are admitted to ICU on Principal Procedure Date or PACU Discharge Date
- Patient expired on Principal Procedure Date
- Patient expired on PACU Discharge Date
- Patient left AMA on PACU Discharge Date
- Documented contraindication by physician/APN/PA/nurse/physical therapist/occupational therapist for not ambulating on day of surgery (e.g. nerve block has not worn off, hypotensive upon standing, patient is vomiting)
- Admission Date
- Birthdate
- Discharge Date
- Discharge Disposition
- ICD-10-CM Other Diagnosis Codes
- ICD-10-CM Principal Diagnosis Code
- ICD-10-PCS Other Procedure Codes
- ICD-10-PCS Principal Procedure Code
- ICD-10-PCS Principal Procedure Date
- PACU Discharge Date
- Postoperative ICU Admit or Transfer
- Reason for Not Ambulating the Day of Surgery
- 1Guerra ML, Singh PJ, Taylor NF. Early mobilization of patients who have had a hip or knee joint replacement reduces length of stay in hospital: A systematic review. Clin Rehabil. 2014 Dec 1.
- 2Tayrose G, Newman D, Slover J, Jaffe F, Hunter T, Bosco J. Rapid Mobilization Decreases Length of Stay in Joint Replacement Patients. Bulletin of the Hospital for Joint Diseases 2013;71(3):222-6.
- AAOS Guidelines on Preventing Venous Thromboembolic Disease in Patients Undergoing Elective Hip and Knee Arthroplasty”, AAOS.
- Premier-IHI Integrated Care Pathway for Total Joint Arthroplasty (April 2013)
- Soohoo NF, Lieberman JR, et al. Development of Quality of Care Indicators for Patients Undergoing THR/TKR. BMJ Qual Saf 2011;20:153-157
- Larsen K, Sorensen, O, Hansen T, Thomsen P, Soballe K. Accelerated perioperative care and rehabilitation intervention for hip and knee replacement is effective: A randomized clinical trial involving 87 patients with 3 months of followup. Acta Orthopaedica 79:2, 149-159.
- den Hertog A, Gliesche K, Timm J, Mühlbauer B, Zebrowski. Pathway-controlled fast-track rehabilitation after total knee arthroplasty: a randomized prospective clinical study evaluating the recovery pattern, drug consumption, and length of stay. Arch Orthop Trauma Surg. 2012 Aug; 132(8):1153-63.
- Smith TO, McCabe C, Lister Set al Rehabilitation implications during the development of the Norwich Enhanced Recovery Programme (NERP) for patients following total knee and total hip arthroplasty. Orthop Traumatol Surg Res 2012; 98: 499–505.
- Raut S, Mertes SC, Muniz-Terrera G, Khanduja V. Factors associated with prolonged length of stay following a total knee replacement in patients aged over 75. Int Orthop 2012; 36: 1,601–1,608.
- Raphael M, Jaeger M, van Vlymen J. Easily adoptable total joint arthroplasty program allows discharge home in two days. Can J Anaesth 2011; 58: 902–910
- Pua YH, Ong PH. Association of early ambulation with length of stay and costs in total knee arthroplasty: retrospective cohort study. Am J Phys Med Rehabil 2014; 93:962-970.
- Chen AF, Stewart MK, Heyl AE, Klatt BA. Effect of Immediate Postoperative Physical Therapy on Length of Stay for Total Joint Arthroplasty Patients. The Journal of Arthroplasty 2012;Vol.27 No. 6
- Wellman SS, Murphy AC, Gulcynski D, Murphy SB. Implementation of an accelerated mobilization protocol following primary total hip arthroplasty: impact on length of stay and disposition. Current Reviews in Musculoskeletal Medicine Volume 4(3); 2011 Sep
- Renkawitz T, Rieder T, Handel M. Comparison of two accelerated clinical pathways – after total knee replacement how fast can we really go? Clinical Rehabilitation 2010; 24:230-239
- Labraca,NS, Castro-Sanchez,AM, Mataran-Penarrocha,G, Arroyo-Morales,M, Sanchez-Joya,M, Moreno-Lorenzo C. Benefits of starting rehabilitation within 24 hours of primary total knee arthroplasty: randomized clinical trial. Clin Rehabil. 2011/25(6):557-566
- Surgical Management of Osteoarthritis of the Knee Evidence-Based Clinical Practice Guideline. Adopted by the American Academy of Orthopaedic Surgeons Board of Directors, 12/4/2015.
CPT® only copyright 2019 American Medical Association. All rights reserved. CPT® is a registered trademark of the American Medical Association.
You, your employees and agents are authorized to use CPT® only as contained in The Joint Commission performance measures solely for your own personal use in directly participating in healthcare programs administered by The Joint Commission. You acknowledge that the American Medical Association (“AMA”) holds all copyright, trademark and other rights in CPT®.
Any use not authorized herein is prohibited, including by way of illustration and not by way of limitation, making copies of CPT® for resale and/or license, transferring copies of CPT® to any party not bound by this Agreement, creating any modified or derivative work of CPT®, or making any commercial use of CPT®. License to use CPT® for any use not authorized herein must be obtained through the American Medical Association, Intellectual Property Services, AMA Plaza, 330 North Wabash Avenue, Suite 39300, Chicago, Illinois 60611-5885. Applications are available at the American Medical Association Web site, www.ama- assn.org/go/cpt.
U.S. Government Rights This product includes CPT® which is commercial technical data, which was developed exclusively at private expense by the American Medical Association, 330 North Wabash Avenue, Chicago, Illinois 60611. The American Medical Association does not agree to license CPT® to the Federal Government based on the license in FAR 52.227-14 (Data Rights - General) and DFARS 252.227-7015 (Technical Data - Commercial Items) or any other license provision. The American Medical Association reserves all rights to approve any license with any Federal agency.
Disclaimer of Warranties and Liabilities. CPT® is provided “as is” without warranty of any kind, either expressed or implied, including but not limited to the implied warranties of merchantability and fitness for a particular purpose. Fee schedules, relative value units, conversion factors and/or related components are not assigned by the AMA, are not part of CPT®, and the (AMA is not recommending their use. The AMA does not directly or indirectly practice medicine or dispense medical services. The responsibility for the content of this product is with The Joint Commission, and no endorsement by the AMA is intended or implied. The AMA disclaims responsibility for any consequences or liability attributable to or related to any use, non-use, or interpretation of information contained or not contained in this product.
This Agreement will terminate upon notice if you violate its terms. The AMA is a third party beneficiary to this Agreement.
Should the foregoing terms and conditions be acceptable to you, please indicate your agreement and acceptance by clicking below on the button labeled “accept”.