Measure Information Form
Version 2019A
Measure Information Form
A single course of corticosteroids should be given at 24 0/7 to 33 6/7 weeks gestation (NIH, 2000). A Cochrane meta-analysis reinforces the beneficial effect of this therapy regardless of membrane status and further concludes for all preterm deliveries the single course of corticosteroids should be routinely administered (Roberts & Dalziel, 2006).
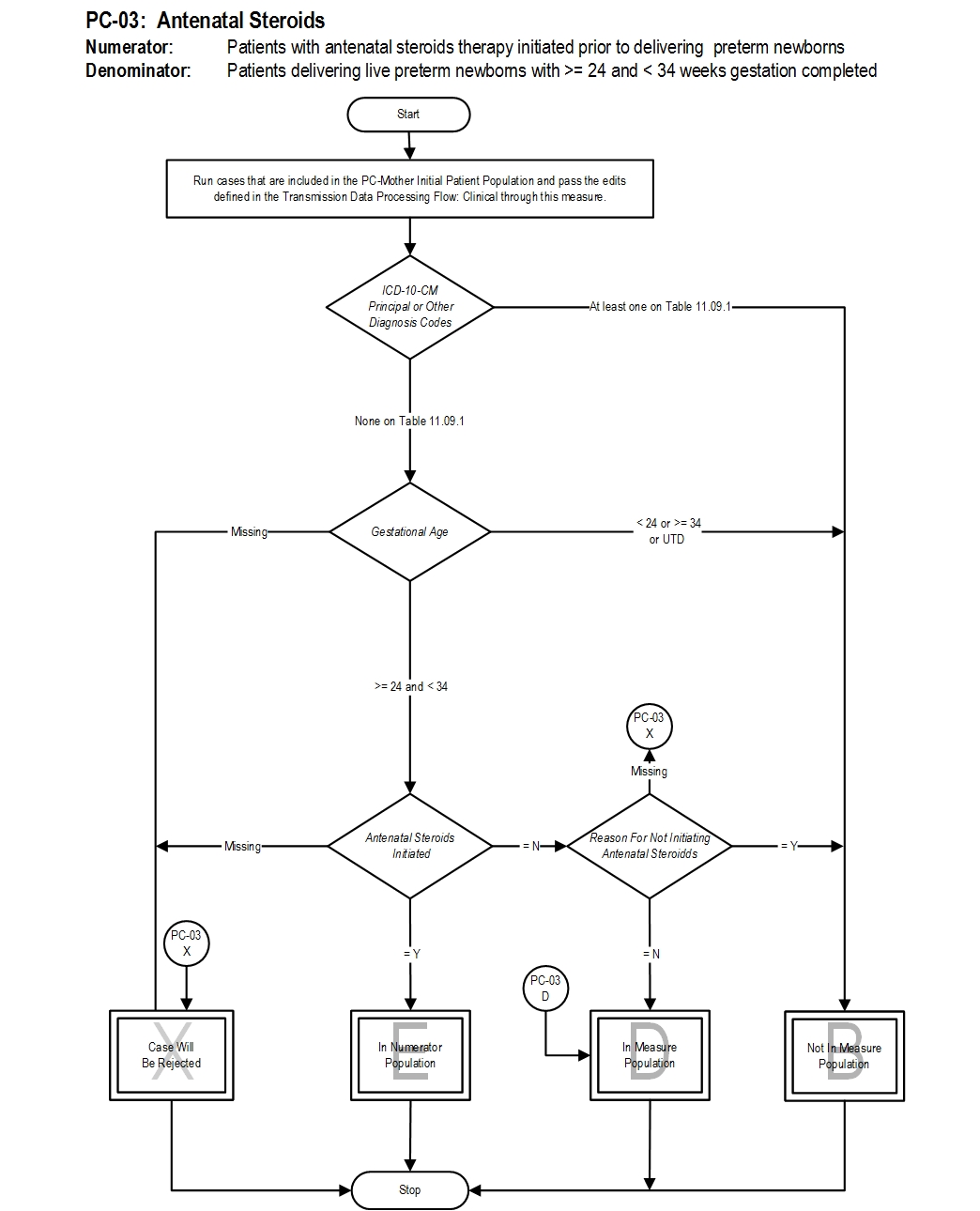
Type Of Measure: Process Improvement Noted As: Increase in the rateIncluded Populations: Antenatal steroids initiated (refer to Appendix C, Table 11.0, antenatal steroid medications) Excluded Populations: None Data Elements:Denominator Statement: Patients delivering live preterm newborns with >=24 and <34 weeks gestation completed
Included Populations:Risk Adjustment: No. Data Collection Approach: Retrospective data sources for required data elements include administrative data and medical records. Data are collected as weeks and days of gestation, but the vendor converts to total days of gestation for data transmission and measure calculation. Data Accuracy: Variation may exist in the assignment of ICD-10 codes; therefore, coding practices may require evaluation to ensure consistency. Measure Analysis Suggestions: In order to identify areas for improvement in antenatal steroid administration rates, hospitals may wish to review documentation for reasons. Education efforts can be targeted based on the specific reasons identified. Sampling: Yes. For additional information see the Sampling Section. Data Reported As: Aggregate rate generated from count data reported as a proportion. Selected References:Excluded Populations:
- ICD-10-PCS Principal Procedure Code or ICD-10-PCS Other Procedure Codes for delivery as defined in Appendix A, Table 11.01.1
Data Elements:
- Less than 8 years of age
- Greater than or equal to 65 years of age
- Length of Stay >120 days
- Documented Reason for Not Initiating Antenatal Steroids
- ICD-10-CM Principal Diagnosis Code or ICD-10-CM Other Diagnosis Codes for fetal demise as defined in Appendix A, Table 11.09.1
- Gestational Age < 24 or >= 34 weeks or UTD
- American College of Obstetricians and Gynecologists. (ACOG). (2013). Practice Bulletin: Clinical Management Guidelines for Obstetrician-Gynecologists for Premature rupture of membranes.
- Lockwood, C.J., ed. & Lemons, J.A., ed. (2007). Guidelines for Perinatal Care, Sixth Edition, American Academy of Pediatrics and the American College of Obstetricians and Gynecologists, ISBN 978-1-58110-270-3; ISBN 978-1-932328-36-3, pp. 178-181.
- NIH Consensus Development Conference Statement: The Effect of Corticosteroids for Fetal Maturation on Perinatal Outcomes. February 28-March 2, 1994.
- NIH Consensus Statement: Antenatal corticosteroids revisited: repeat courses.2000. 17(2)1-18.
- Roberts, D. & Dalziel, S.R. (2010) Antenatal corticosteroids for accelerating fetal lung maturation for women at risk of preterm birth (Review). The Cochrane Collaboration. Issue 9.
Providence St Vincent's Hospital/Council of Women and Infant's Specialty Hospitals
CPT® only copyright 2019 American Medical Association. All rights reserved. CPT® is a registered trademark of the American Medical Association.
You, your employees and agents are authorized to use CPT® only as contained in The Joint Commission performance measures solely for your own personal use in directly participating in healthcare programs administered by The Joint Commission. You acknowledge that the American Medical Association (“AMA”) holds all copyright, trademark and other rights in CPT®.
Any use not authorized herein is prohibited, including by way of illustration and not by way of limitation, making copies of CPT® for resale and/or license, transferring copies of CPT® to any party not bound by this Agreement, creating any modified or derivative work of CPT®, or making any commercial use of CPT®. License to use CPT® for any use not authorized herein must be obtained through the American Medical Association, Intellectual Property Services, AMA Plaza, 330 North Wabash Avenue, Suite 39300, Chicago, Illinois 60611-5885. Applications are available at the American Medical Association Web site, www.ama- assn.org/go/cpt.
U.S. Government Rights This product includes CPT® which is commercial technical data, which was developed exclusively at private expense by the American Medical Association, 330 North Wabash Avenue, Chicago, Illinois 60611. The American Medical Association does not agree to license CPT® to the Federal Government based on the license in FAR 52.227-14 (Data Rights - General) and DFARS 252.227-7015 (Technical Data - Commercial Items) or any other license provision. The American Medical Association reserves all rights to approve any license with any Federal agency.
Disclaimer of Warranties and Liabilities. CPT® is provided “as is” without warranty of any kind, either expressed or implied, including but not limited to the implied warranties of merchantability and fitness for a particular purpose. Fee schedules, relative value units, conversion factors and/or related components are not assigned by the AMA, are not part of CPT®, and the (AMA is not recommending their use. The AMA does not directly or indirectly practice medicine or dispense medical services. The responsibility for the content of this product is with The Joint Commission, and no endorsement by the AMA is intended or implied. The AMA disclaims responsibility for any consequences or liability attributable to or related to any use, non-use, or interpretation of information contained or not contained in this product.
This Agreement will terminate upon notice if you violate its terms. The AMA is a third party beneficiary to this Agreement.
Should the foregoing terms and conditions be acceptable to you, please indicate your agreement and acceptance by clicking below on the button labeled “accept”.