Measure Information Form
Version 2019A
Measure Information Form
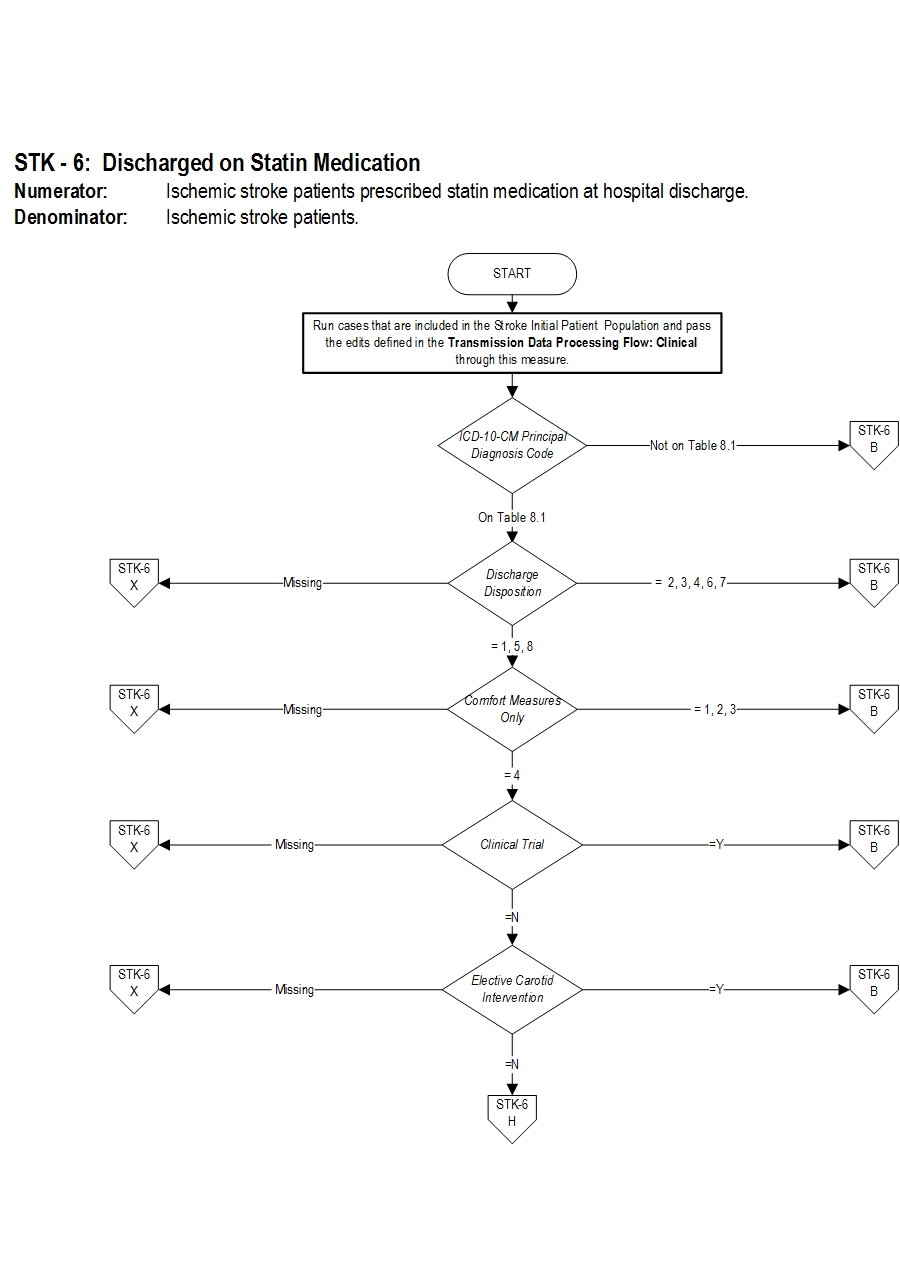
Included Populations: Not applicable Excluded Populations: None Data Elements:Denominator Statement: Ischemic stroke patients
Included Populations: Discharges with an ICD-10-CM Principal Diagnosis Code for ischemic stroke as defined in Appendix A, Table 8.1 Excluded Populations:Risk Adjustment: No. Data Collection Approach: Retrospective data sources for required data elements include administrative data and medical record documents. Some hospitals may prefer to gather data concurrently by identifying patients in the population of interest. This approach provides opportunities for improvement at the point of care/service. However, complete documentation includes the principal or other ICD-10 diagnosis and procedure codes, which require retrospective data entry. Data Accuracy: Variation may exist in the assignment of ICD-10 codes; therefore, coding practices may require evaluation to ensure consistency. Measure Analysis Suggestions: None Sampling: Yes. Please refer to the measure set specific sampling requirements and for additional information see the Population and Sampling Specifications section Data Reported As: Aggregate rate generated from count data reported as a proportion. Selected References:Data Elements:
- Patients less than 18 years of age
- Patients who have a Length of Stay greater than 120 days
- Patients with Comfort Measures Only documented
- Patients enrolled in clinical trials
- Patients admitted for Elective Carotid Intervention
- Patients discharged to another hospital
- Patients who left against medical advice
- Patients who expired
- Patients discharged to home for hospice care
- Patients discharged to a health care facility for hospice care
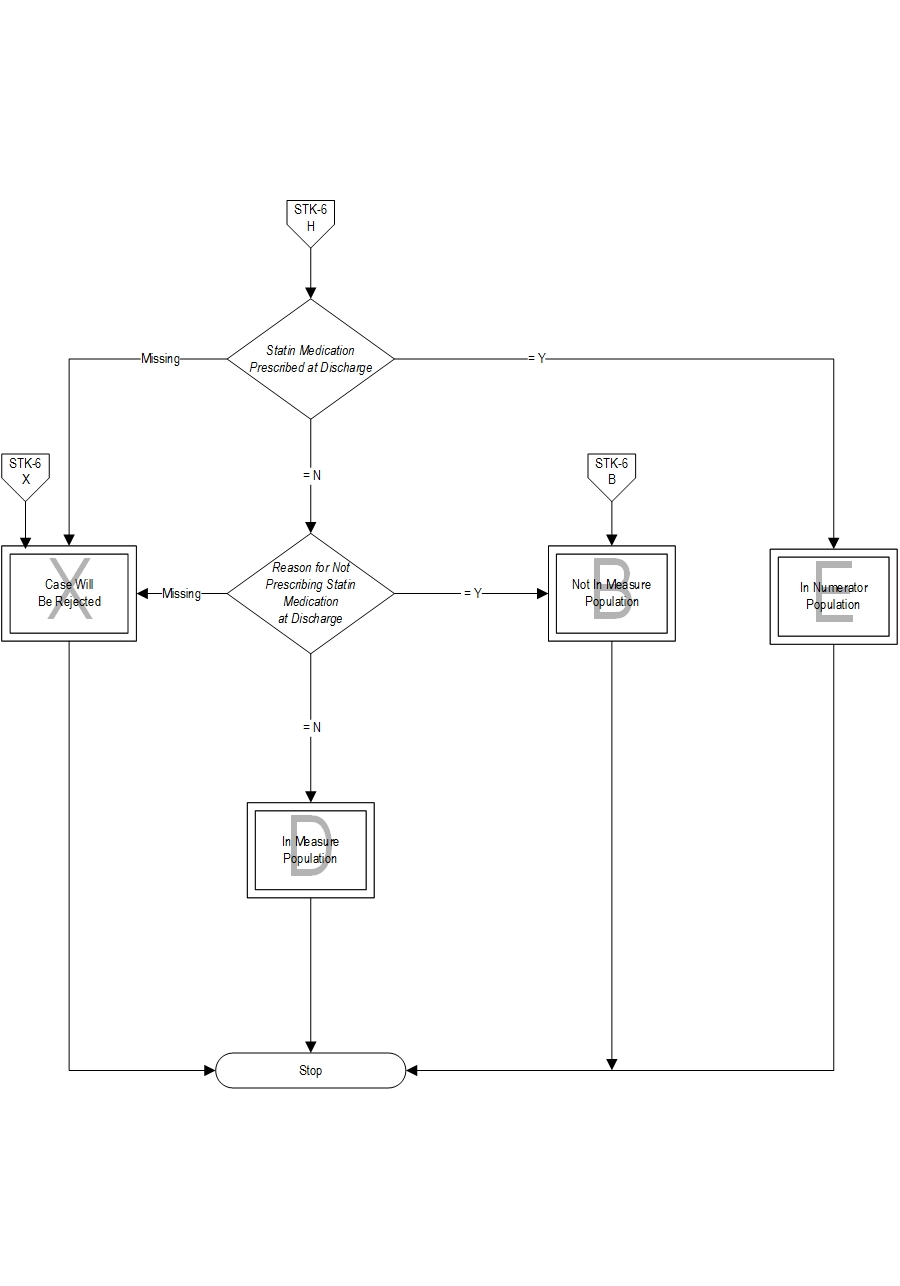
- Patients with a Reason for Not Prescribing Statin Medication at Discharge
- Amarenco, P., J. Bogousslavsky, A. Callahan, 3rd, L. B. Goldstein, M. Hennerici, A. E. Rudolph, H. Sillesen, et al. "High-Dose Atorvastatin after Stroke or Transient Ischemic Attack." [In eng]. N Engl J Med 355, no. 6 (Aug 10 2006): 549-59.
- Biffi, A., W. J. Devan, C. D. Anderson, L. Cortellini, K. L. Furie, J. Rosand, and N. S. Rost. "Statin Treatment and Functional Outcome after Ischemic Stroke: Case-Control and Meta-Analysis." [In eng]. Stroke 42, no. 5 (May 2011): 1314-9.
- Centers for Disease Control and Prevention. "Prevalence and Most Common Causes of Disability among Adults--United States, 2005." [In eng]. MMWR Morb Mortal Wkly Rep 58, no. 16 (May 1 2009): 421-6.
- Chan, P. S., B. K. Nallamothu, H. S. Gurm, R. A. Hayward, and S. Vijan. "Incremental Benefit and Cost-Effectiveness of High-Dose Statin Therapy in High-Risk Patients with Coronary Artery Disease." [In eng]. Circulation 115, no. 18 (May 8 2007): 2398-409.
- Culver, A. L., I. S. Ockene, R. Balasubramanian, B. C. Olendzki, D. M. Sepavich, J. Wactawski-Wende, J. E. Manson, et al. "Statin Use and Risk of Diabetes Mellitus in Postmenopausal Women in the Women's Health Initiative." [In eng]. Arch Intern Med 172, no. 2 (Jan 23 2012): 144-52.
- Feher, A., G. Pusch, K. Koltai, A. Tibold, B. Gasztonyi, L. Szapary, and G. Feher. "Statintherapy in the Primary and the Secondary Prevention of Ischaemic Cerebrovascular Diseases." [In eng]. Int J Cardiol 148, no. 2 (Apr 14 2011): 131-8.
- Grundy, S. M., J. I. Cleeman, C. N. Merz, H. B. Brewer, Jr., L. T. Clark, D. B. Hunninghake, R. C. Pasternak, et al. "Implications of Recent Clinical Trials for the National Cholesterol Education Program Adult Treatment Panel III Guidelines." [In eng]. Circulation 110, no. 2 (Jul 13 2004): 227-39.
- Grundy, S. M., Stone, N. J., Bailey, A. L., Beam, C., Birtcher, K. K., Blumenthal, R. S., et. al. “Guideline on the Management of Blood Cholesterol: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines." [In eng]. Journal of the American College of Cardiology (2018), doi: https://doi.org/10.1016/j.jacc.2018.11.003.
- Kernan, W.N., B. Ovbiagele, H. R. Black, D. M. Bravata, M. I. Chimowitz, M. D. Ezekowitz, M. C. Fang, M. Fisher, K. L. Furie, D. V. Heck, S. C. Johnston, S. E. Kasner, S. J. Kittner, P. H. Mitchell, M. W. Rich, D. Richardson, L. H. Schwamm, J. A. Wilson. “Guidelines for the Prevention of Stroke in Patients with Stroke and Transient Ischemic Attack: A Guideline for Healthcare Professionals from the American Heart Association/American Stroke Association.” [in eng.] Stroke 45, no. 7 (May 2014): 2160-223.
- Kostis, W. J., J. Q. Cheng, J. M. Dobrzynski, J. Cabrera, and J. B. Kostis. "Meta-Analysis of Statin Effects in Women Versus Men." [In eng]. J Am Coll Cardiol 59, no. 6 (Feb 7 2012): 572-82.
- Lazar, L. D., M. J. Pletcher, P. G. Coxson, K. Bibbins-Domingo, and L. Goldman. "Cost-Effectiveness of Statin Therapy for Primary Prevention in a Low-Cost Statin Era." [In eng]. Circulation 124, no. 2 (Jul 12 2011): 146-53.
- Mitka, M. "Some Question Use of Statins to Reduce Cardiovascular Risks in Healthy Women." [In eng]. JAMA 307, no. 9 (Mar 7 2012): 893-4.
- National Cholesterol Education Program Expert Panel on Detection, Evaluation, and Adults Treatment of High Blood Cholesterol in Adults."Third Report of the National Cholesterol Education Program (Ncep) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel Iii) Final Report." [In eng]. Circulation 106, no. 25 (Dec 17 2002): 3143-421.
- Powers WJ, Rabinstein AA, Ackerson T, Adeoye OM, Bambakidis NC, Becker K, Biller J, et al; on behalf of the American Heart Association Stroke Council. 2018 Guidelines for the Early Management of Patients with Acute Ischemic Stroke: A Guideline for Healthcare Professionals From the American Heart Association/American Stroke Association. Stroke. 2018 Jan;49:e44, e47.
- Rodriguez-Yanez, M., J. Agulla, R. Rodriguez-Gonzalez, T. Sobrino, and J. Castillo. "Statins and Stroke." [In eng]. Ther Adv Cardiovasc Dis 2, no. 3 (Jun 2008): 157-66.
- Roger, V. L., A. S. Go, D. M. Lloyd-Jones, E. J. Benjamin, J. D. Berry, W. B. Borden, D. M. Bravata, et al. "Heart Disease and Stroke Statistics--2012 Update: A Report from the American Heart Association." [In eng]. Circulation 125, no. 1 (Jan 3 2012): e2-e220.
- Schellinger, P. D., R. N. Bryan, L. R. Caplan, J. A. Detre, R. R. Edelman, C. Jaigobin, C. S. Kidwell, et al. "Evidence-Based Guideline: The Role of Diffusion and Perfusion MRI for the Diagnosis of Acute Ischemic Stroke: Report of the Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology." [In eng]. Neurology 75, no. 2 (Jul 13 2010): 177-85.
- Squizzato, A., E. Romualdi, F. Dentali, and W. Ageno. "Statins for Acute Ischemic Stroke." [In eng]. Cochrane Database Syst Rev, no. 8 (2011): CD007551.
- Stone NJ, Robinson J, Lichtenstein AH, Noel Bairey Merz C, Blum CB, Eckel RH, Goldberg AC, Gordon D, Levy D, Lloyd-Jones DM, McBride P, Schwartz JS, Shero Jr, ST, Smith SC, Watson K, Wilson PWF. “Guideline on the Treatment of Blood Cholesterol to Reduce Atherosclerotic Cardiovascular Disease in Adults: A Report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. [In eng]. Circulation 11, (Nov 2013): 1-84.
- Van Dis, F. J., L. M. Keilson, C. A. Rundell, and M. W. Rawstron. "Direct Measurement of Serum Low-Density Lipoprotein Cholesterol in Patients with Acute Myocardial Infarction on Admission to the Emergency Room." [In eng]. Am J Cardiol 77, no. 14 (Jun 1 1996): 1232-4.
- Weiss, R., M. Harder, and J. Rowe. "The Relationship between Nonfasting and Fasting Lipid Measurements in Patients with or without Type 2 Diabetes Mellitus Receiving Treatment with 3-Hydroxy-3-Methylglutaryl-Coenzyme a Reductase Inhibitors." [In eng]. Clin Ther 25, no. 5 (May 2003): 1490-7.
CPT® only copyright 2019 American Medical Association. All rights reserved. CPT® is a registered trademark of the American Medical Association.
You, your employees and agents are authorized to use CPT® only as contained in The Joint Commission performance measures solely for your own personal use in directly participating in healthcare programs administered by The Joint Commission. You acknowledge that the American Medical Association (“AMA”) holds all copyright, trademark and other rights in CPT®.
Any use not authorized herein is prohibited, including by way of illustration and not by way of limitation, making copies of CPT® for resale and/or license, transferring copies of CPT® to any party not bound by this Agreement, creating any modified or derivative work of CPT®, or making any commercial use of CPT®. License to use CPT® for any use not authorized herein must be obtained through the American Medical Association, Intellectual Property Services, AMA Plaza, 330 North Wabash Avenue, Suite 39300, Chicago, Illinois 60611-5885. Applications are available at the American Medical Association Web site, www.ama- assn.org/go/cpt.
U.S. Government Rights This product includes CPT® which is commercial technical data, which was developed exclusively at private expense by the American Medical Association, 330 North Wabash Avenue, Chicago, Illinois 60611. The American Medical Association does not agree to license CPT® to the Federal Government based on the license in FAR 52.227-14 (Data Rights - General) and DFARS 252.227-7015 (Technical Data - Commercial Items) or any other license provision. The American Medical Association reserves all rights to approve any license with any Federal agency.
Disclaimer of Warranties and Liabilities. CPT® is provided “as is” without warranty of any kind, either expressed or implied, including but not limited to the implied warranties of merchantability and fitness for a particular purpose. Fee schedules, relative value units, conversion factors and/or related components are not assigned by the AMA, are not part of CPT®, and the (AMA is not recommending their use. The AMA does not directly or indirectly practice medicine or dispense medical services. The responsibility for the content of this product is with The Joint Commission, and no endorsement by the AMA is intended or implied. The AMA disclaims responsibility for any consequences or liability attributable to or related to any use, non-use, or interpretation of information contained or not contained in this product.
This Agreement will terminate upon notice if you violate its terms. The AMA is a third party beneficiary to this Agreement.
Should the foregoing terms and conditions be acceptable to you, please indicate your agreement and acceptance by clicking below on the button labeled “accept”.