Release Notes:
Measure Information Form
Version 2018B1
Measure Information Form
Version 2018B1
Measure Information Form
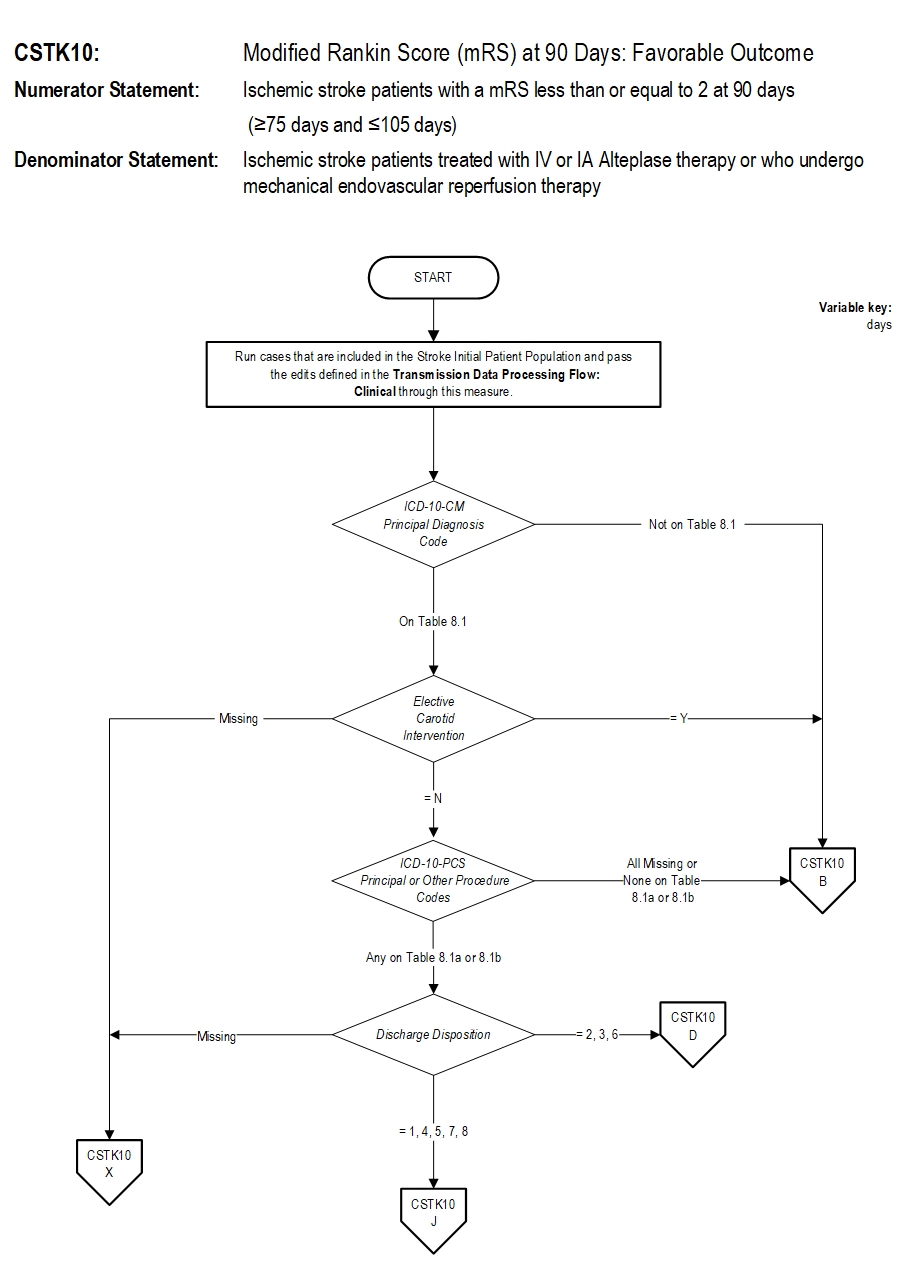
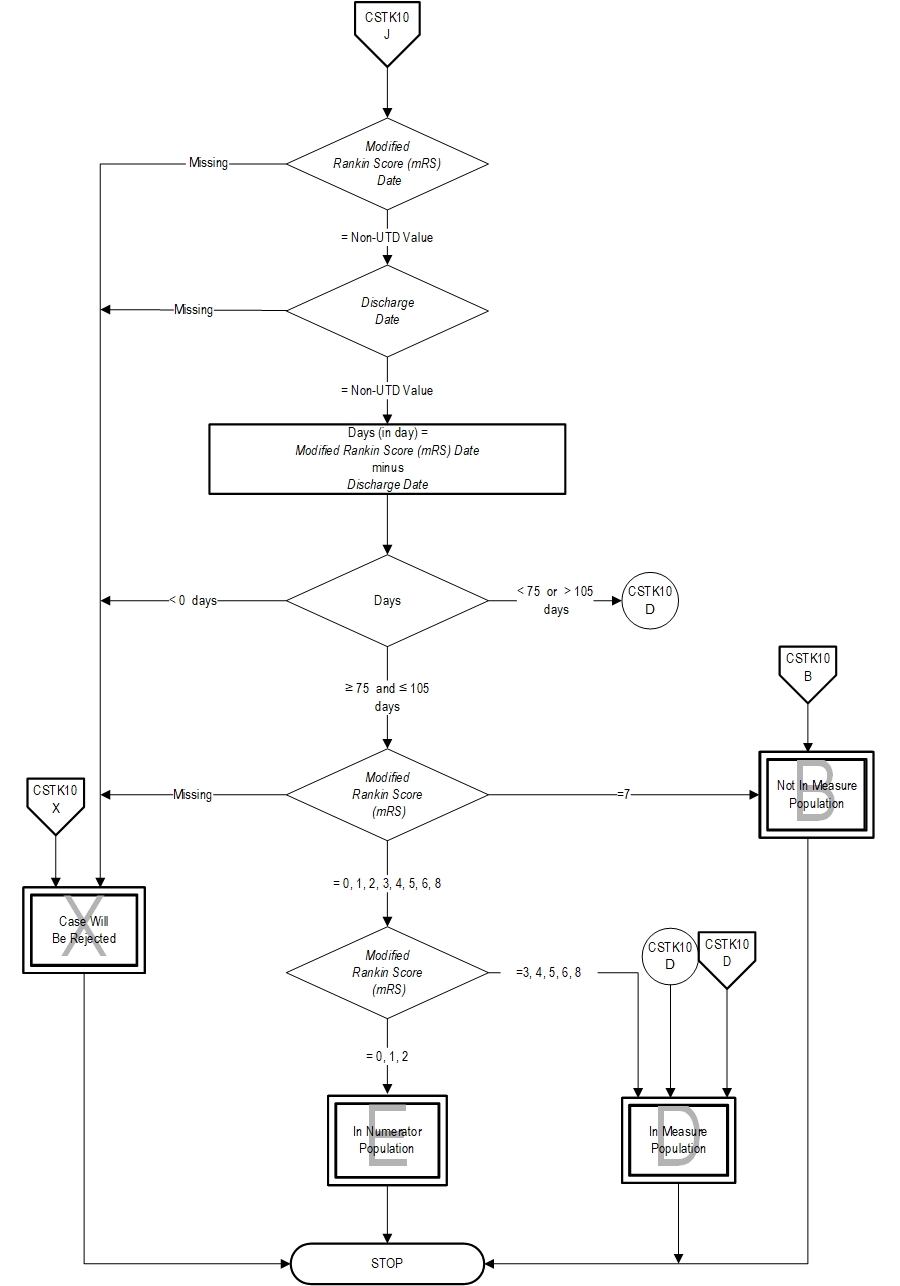
Numerator Statement: Ischemic stroke patients with a mRS less than or equal to 2 at 90 days (≥75 days and ≤105 days)
Included Populations: As above Excluded Populations: None Data Elements:Denominator Statement: Ischemic stroke patients treated with IV or IA alteplase therapy or who undergo mechanical endovascular reperfusion therapy
Included Populations:Risk Adjustment: Yes. This section has been moved to the ORYX Risk Adjustment Guide. This guide is available to the public on the Joint Commission's website and, in addition, it is available to performance measurement systems via the Joint Commission's extranet site for measurement systems (PET).Excluded Populations:
- Discharges with ICD-10-CM Principal Diagnosis Code for ischemic stroke as defined in Appendix A, Table 8.1 for ICD-10 codes, AND
- Patients with documented thrombolytic (IV or IA alteplase) therapy (ICD-10-PCS Principal or Other Procedure Codes as defined in Appendix A, Table 8.1a for ICD-10 codes), OR
- Patients with documented Mechanical Endovascular Reperfusion Therapy (ICD-10-PCS Principal or Other Procedure Codes as defined in Appendix A, Table 8.1b for ICD-10 codes)
Data Elements:
- Patients less than 18 years of age
- Patients who have a Length of Stay > 120 days
- Patients admitted for Elective Carotid Intervention
- Patients and their caregivers who cannot be contacted via phone or in-person at 90 days
Data Elements:Data Collection Approach: Retrospective data sources for required data elements include administrative data and, if applicable, medical record documents. Data Accuracy: Variation may exist in the assignment of ICD-10 codes; therefore, coding practices may require evaluation to ensure consistency. Measure Analysis Suggestions: None Sampling: Yes. Please refer to the measure set specific sampling requirements and for additional information see the Population and Sampling Specifications section. Data Reported As: Aggregate rate generated from count data reported as a proportion. Selected References: 1. Adams HP, del Zoppo G, Alberts MJ, Bhatt DL, Brass L, Furlan A, Grubb RL, Higashida RT, Jauch EC, Kidwell C, Lyden PD, Morgenstern LB, Qureshi AI, Rosenwasser RH, Scott PA, Wijdicks E. Guidelines for the Early Management of Adults with Ischemic Stroke: A Guideline From the American Heart Association/American Stroke Association Stroke Council, Clinical Cardiology Council, Cardiovascular Radiology and Intervention Council, and the Atherosclerotic Peripheral Vascular Disease and Quality of Care Outcomes in Research Interdisciplinary Working Groups. Stroke. 2007;38:1675-1678. 2. Banks JL, Marotta CA. Outcomes validity and reliability of the modified Rankin scale: implications for stroke clinical trials: a literature review and synthesis. Stroke. 2007:38:2262-2269. 3. Bruno A, Shah N, Lin C, Close B, Hess DC, Davis K, Baute V, Switzer JA, Waller JL, Nichols FT. Simplified modified Rankin scale questionnaire: reproducibility over the telephone and validation with quality of life. Stroke. 2011;42:2276-2279. 4. Campbell BCV, Mitchell PJ, Kleinig TJ, Dewey HM, Churilov L, Yassi N, et. al. Endovascular therapy for ischemic stroke with perfusion-imaging selection. NEJM. 2015 Mar;372(11): 1009-17. 5. Demchuk AM, Goyal M, Monon BK, Eesa M, Ryckborst KJ, Kamal N, et. al. Endovascular treatment for Small Core and Anterior circulation Proximal occlusion with Emphasis on minimizing CT to recanalization times (ESCAPE) trial: methodology. Int J Stroke. 2015 Apr;10(3): 429-38. 6. Jauch EC, Saver JL, Adams HP Jr, Bruno A, Connors JJ, Demaerschalk BM, Khatri P, et al. Guidelines for the early management of patients with acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2013;44:32-36. 7. Leifer D, Bravata DM, Connors JJ III, Hinchey JA, Jauch EC, Johnston SC, Latchaw R, Likosky W, Ogilvy C, Qureshi AI, Summers D, Sung GY, Williams LS, Zorowitz R, on behalf of the American Heart Association Special Writing Group of the Stroke Council, Atherosclerotic Peripheral Vascular Disease Working Group and Council on Cardiovascular Surgery and Anesthesia, and Council on Cardiovascular Nursing. Metrics for measuring quality of care in comprehensive stroke centers: detailed follow-up to Brain Attack Coalition comprehensive stroke center recommendations: a statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2011;42:857. 8. Powers WJ, Rabinstein AA, Ackerson T, Adeoye OM, Bambakidis NC, Becker K, Biller J, et al; on behalf of the American Heart Association Stroke Council. 2018 Guidelines for the Early Management of Patients with Acute Ischemic Stroke: A Guideline for Healthcare Professionals From the American Heart Association/American Stroke Association. Stroke. 2018 Jan;49:e10-e11. 9. Quinn TJ, Dawson J, Walters MR, Lees KR. Reliability of the modified Rankin scale. Stroke. 2007:38:e144. 10. Rankin J. Cerebral vascular accidents in patients over the age of 60. Scott Med J. 1957;2(5):200-15. 11. Saver JL, Goyal M, Bonafe A, Diener HC, Levy EI, Pereira VM, et. al. Stent-retriever thrombectomy after intravenous t-PA vs. t-PA alone in stroke. NEJM. 2015 Apr: 1-11. 12. Schwamm LH, Holloway RG, Amarenco P. Audebert HJ, Bakas T, Chumbler NR, Handschu R, Jauch EC, Knight WA IV, Levine SR, Mayberg M, Meyer BC, Meyers PM, Skalabrin E, Wechsler LR; American Heart Association Stroke Council; Interdisciplinary Council on Peripheral Vascular Disease. A review of the evidence for the use of telemedicine within stroke systems of care: a scientific statement for the American Heart Association/American Stroke Association. Stroke. 2009;40:2616-2634. 13. The National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group. Tissue plasminogen activator for acute ischemic stroke. The National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group. New England Journal of Medicine 1995;333:1581-1587. 14. Turk AS, Frei D, Fiorella D, Mocco J, Baxter B, Siddiqui A, et. al. ADAPT FAST study: a direct aspiration first pass technique for acute stroke thrombectomy. J Neurointerv Surg. 2014 May;694): 260-4. 15. Wilson JT, Hareendran A, Hendry A, Potter J. Bone I, Muir KW. Reliability of the modified Rankin scale across multiple raters: benefits of a structured interview. Stroke. 2005;36:777-781.
- Admission Date
- Hispanic Ethnicity
- Race
- Sex
- Birthdate
- Initial Blood Glucose Value at Hospital Arrival
- Initial NIHSS Score at Hospital Arrival
- Initial Platelet Count at Hospital Arrival
- Initial Blood Pressure at Hospital Arrival
- IV Alteplase Prior to IA or Mechanical Reperfusion Therapy
- ICD-10-CM Other Diagnosis Codes
- Pre-Stroke Modified Rankin Score (mRS)
Measure Algorithm:


Questions? Ask Question to Joint Commission staff
Copyright © 2018 by The Joint Commission.