Release Notes:
Measure Information Form
Version 2018A1
Measure Information Form
Version 2018A1
**NQF-ENDORSED VOLUNTARY CONSENSUS STANDARDS FOR HOSPITAL CARE**
Measure Information Form
Numerator Statement: SAH patients for whom nimodipine treatment was administered within 24 hours of arrival at this hospital.
Risk Adjustment: No.
Data Collection Approach: Retrospective data sources for required data elements include administrative data and medical records.
Data Accuracy: Variation may exist in the assignment of ICD-10 codes; therefore, coding practices may require evaluation to ensure consistency.
Measure Analysis Suggestions: None
Sampling: Yes. Please refer to the measure set specific sampling requirements and for additional information see the Population and Sampling Specifications section.
Data Reported As: Aggregate rate generated from count data reported as a proportion.
Selected References:
1. Adams HP, del Zoppo G, Alberts MJ, Bhatt DL, Brass L, Furlan A, Grubb RL, Higashida RT, Jauch EC, Kidwell C, Lyden PD, Morgenstern LB, Qureshi AI, Rosenwasser RH, Scott PA, Wijdicks E. Guidelines for the Early Management of Adults with Ischemic Stroke: A Guideline From the American Heart Association/American Stroke Association Stroke Council, Clinical Cardiology Council, Cardiovascular Radiology and Intervention Council, and the Atherosclerotic Peripheral Vascular Disease and Quality of Care Outcomes in Research Interdisciplinary Working Groups. Stroke. 2007;38:1686.
2. Allen GS, Ahn HS, Presiosi TJ, Battye R,Boone SC, Cho SN, Kelly DL, Weir BK, Crabbe RA, Lavik PJ, Rosenbloom SB, Dorsey FC, Ingram CR, Mellits DE, Bertsch LA, Boisvert DP, Hundley MB, Johnson RK, Strom JA, Transou CR. Cerebral arterial spasm: a controlled trial of nimodipine in patients with subarachnoid hemorrhage. N Engl J Med. 1983;308:619-624.
3. Bederson JB, Connolly ES Jr, Batjer HH, Dacey RG, Dion JE, Diringer MN, Duldner JE Jr, Harbaugh RE, Patel AB, and Rosenwasser RH. Guidelines for the management of aneurysmal subarachnoid hemorrhage: a statement for healthcare professionals from a Special Writing Group of the Stroke Council, American Heart Association. Stroke. 2009;40:1008-1011.
Included Populations: As above Excluded Populations: None Data Elements:Denominator Statement: SAH patients
Included Populations: Discharges with ICD-10-CM Principal Diagnosis Code for subarachnoid hemorrhage as defined in Appendix A, Table 8.2a for ICD-10 codes. Excluded Populations:Data Elements:
- Patients less than 18 years of age
- Patients who have a Length of Stay > 120 days
- Patients with Comfort Measures Only documented on day of or after hospital arrival
- Patients enrolled in clinical trials
- Patients discharged within 24 hours of arrival at this hospital
4. Connolly ES, Rabinstein AA, Carhuapoma JR, Derdeyn CP, Dion J, Higashida RT, Hoh BL, Kirkness CJ, Naidech AM, Ogilvy CS, Patel AB, Thompson BG, Vespa P. Guidelines for the management of aneurysmal subarachnoid hemorrhage: a guidelines for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2012;43:1-27.
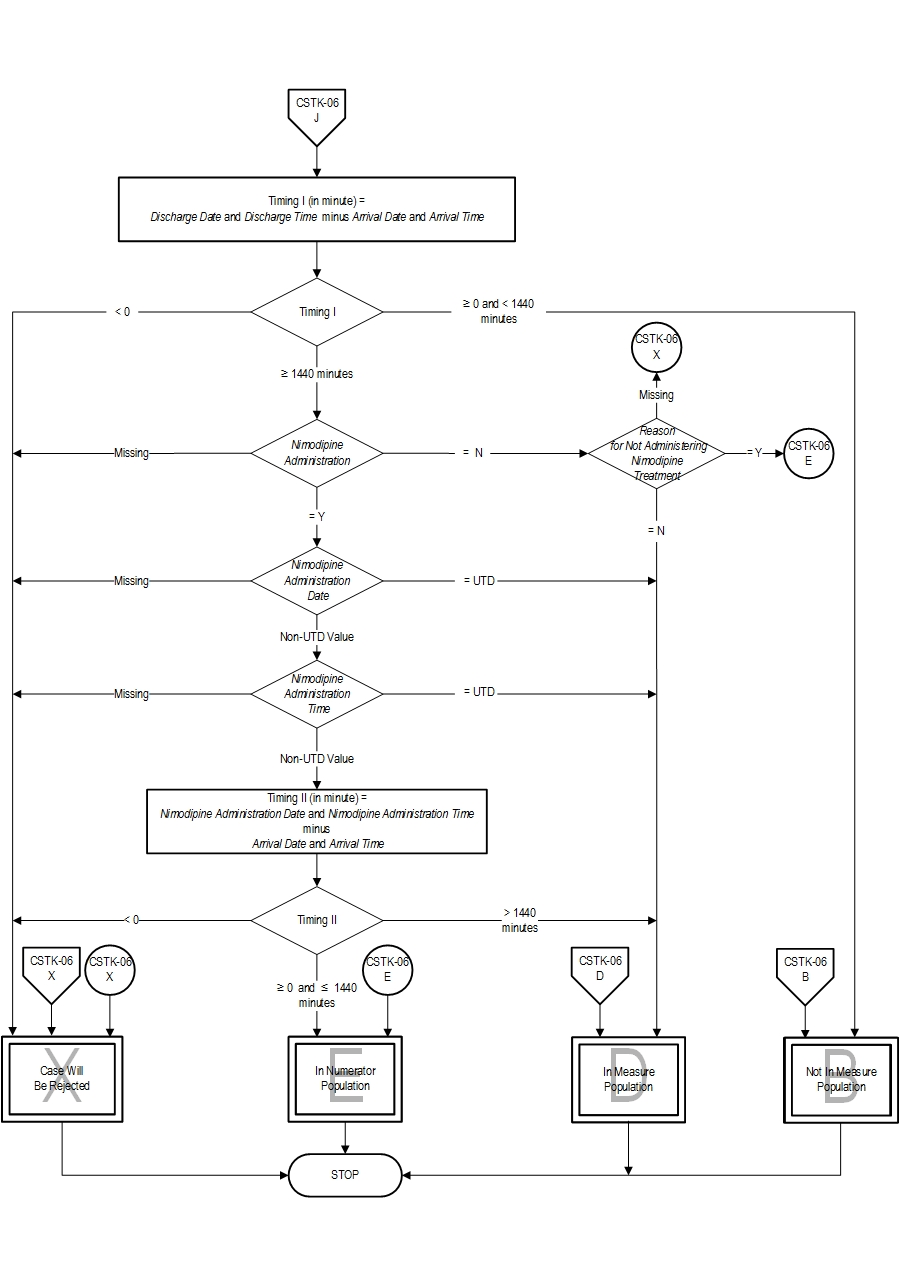
5. Fogelholm R, Palomaki H, Erila T, Rissanen A, Kaste M. Blood pressure, nimodipine, and outcome of ischemic stroke. Acta Neurol Scand. 2004;109:200-204. 6. Haley EC Jr, Kassell NF, Torner JC, Truskowski LL, Germanson TP. A randomized trial of two doses of nicardipine in aneurysmal subarachnoid hemorrhage: a report of the Cooperative Aneurysm Study. J Neurosugr. 1994;80:788-796. 7. Kaste M, Fogelholm R, Erila T, Palomaki H, Murros K, Rissanen A, Sarna S. A randomized, double-blinded, placebo-controlled trial of nimodipine in acute ischemic hemispheric stroke. Stroke. 1994;25:1348-1353. 8. Leifer D, Bravata DM, Connors JJ III, Hinchey JA, Jauch EC, Johnston SC, Latchaw R, Likosky W, Ogilvy C, Qureshi AI, Summers D, Sung GY, Williams LS, Zorowitz R, on behalf of the American Heart Association Special Writing Group of the Stroke Council, Atherosclerotic Peripheral Vascular Disease Working Group and Council on Cardiovascular Surgery and Anesthesia, and Council on Cardiovascular Nursing. Metrics for measuring quality of care in comprehensive stroke centers: detailed follow-up to Brain Attack Coalition comprehensive stroke center recommendations: a statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2011;42:863-864. 9. Mayberg MR, Batjer HH, Dacey R, Diringer M, Haley EC, Heros RC, Sternau LL, Torner J, Adams HP Jr, Feinberg W. Guidelines for the management of aneurysmal subarachnoid hemorrhage: a statement for healthcare professionals from a Special Writing Group of the Stroke Council, American Heart Association. Stroke. 1994;25:2315-2328. 10. Toyota BD. The efficacy of an abbreviated course of nimodipine in patients with good grade aneurysmal subarachnoid hemorrhage. JNeurosurg. 1999;90(2):203-206. 11. Wahlgren NG, Applications/LocalApps.MacMahon DG, Applications/LocalApps.DeKeyser J, Indredavik B, Ryman T. Intravenous Nimodipine West European Stroke Trial (INWEST) of nimodipine in the treatment of acute ischemic stroke. Cerebrovasc Dis. 1994;4:204-210. 12. The American Nimodipine Study Group. Clinical trial of nimodipine in acute ischemic stroke. Stroke. 1992;23:3-8. Measure Algorithm: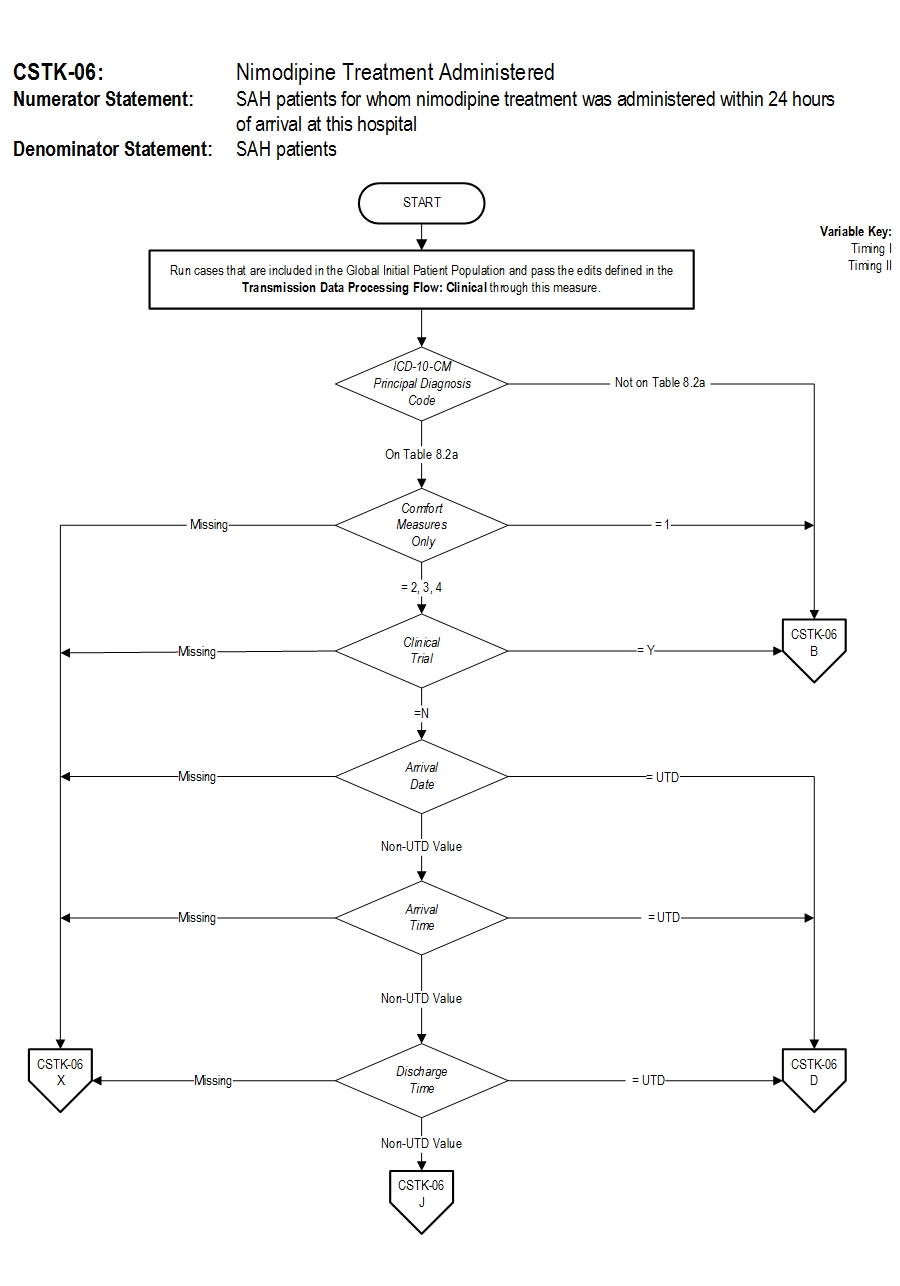
| Related Topics |
Questions? Ask Question to Joint Commission staff
Copyright © 2018 by The Joint Commission.