Release Notes:
Measure Information Form
Version 2016A1
**NQF-ENDORSED VOLUNTARY CONSENSUS STANDARDS FOR HOSPITAL CARE**Measure Information Form
Measure Set: Stroke(STK)
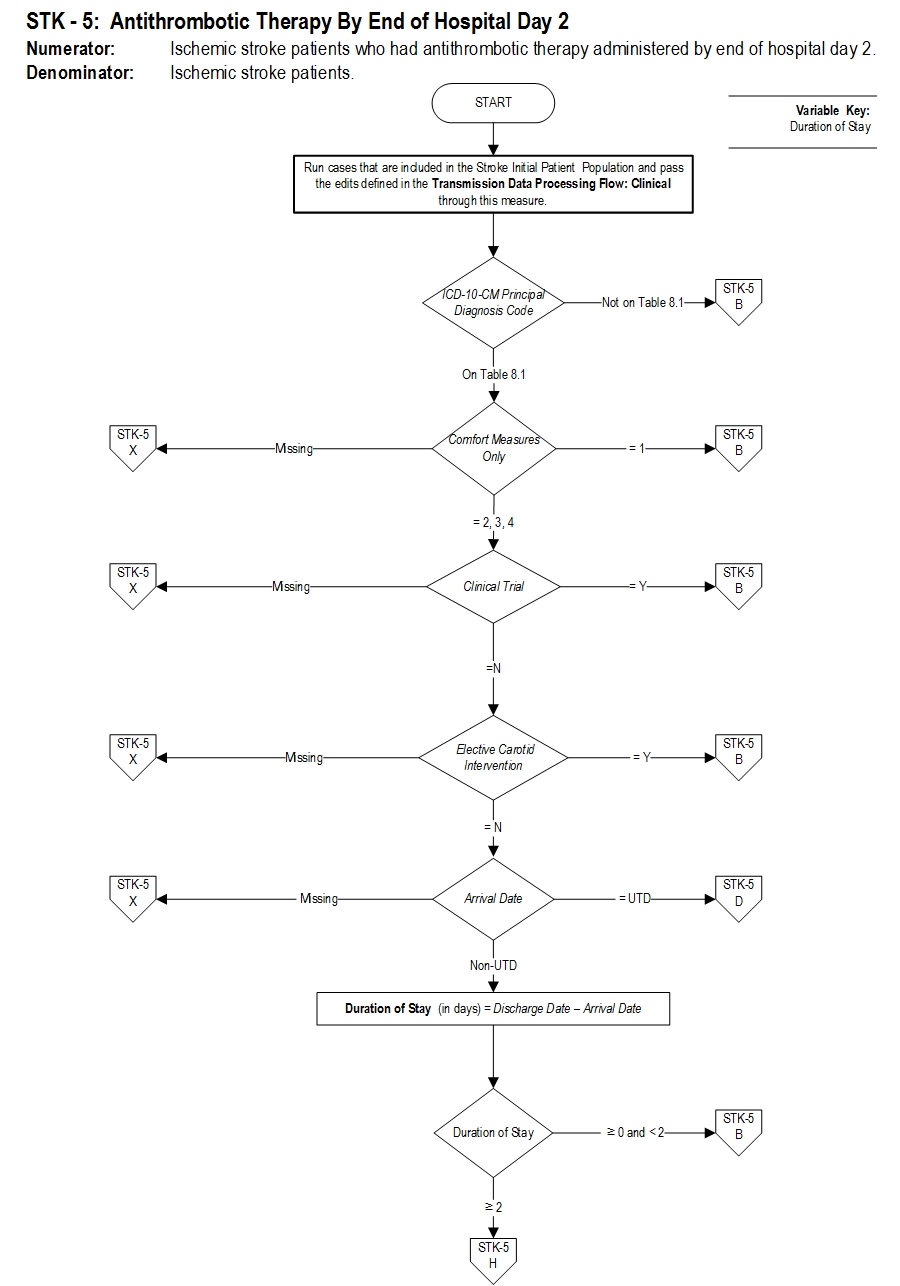
Set Measure ID: STK-5
Performance Measure Name: Antithrombotic Therapy By End of Hospital Day Two
Description: Ischemic stroke patients administered antithrombotic therapy by the end of hospital day 2.
Rationale: The effectiveness of antithrombotic agents in reducing stroke mortality, stroke-related morbidity and recurrence rates has been studied in several large clinical trials. While the use of these agents for patients with acute ischemic stroke and transient ischemic attacks continues to be the subject of study, substantial evidence is available from completed studies. Data at this time suggest that antithrombotic therapy should be administered within 2 days of symptom onset in acute ischemic stroke patients to reduce stroke mortality and morbidity as long as no contraindications exist.
Type of Measure: Process
Improvement Noted As: Increase in the rate
Numerator Statement: Ischemic stroke patients who had antithrombotic therapy administered by end of hospital day 2.
Included Populations: Not applicable
Excluded Populations: None
Data Elements:
Denominator Statement: Ischemic stroke patients.
Included Populations:
Discharges with an ICD-10-CM Principal Diagnosis Code for ischemic stroke as defined in Appendix A, Table 8.1.
Excluded Populations:
- Patients less than 18 years of age
- Patients who have a Duration of Stay less than 2 days
- Patients who have a Length of Stay greater than 120 days
- Patients with Comfort Measures Only documented on day of or day after arrival
- Patients enrolled in clinical trials
- Patients admitted for Elective Carotid Intervention
- Patients discharged prior to the end of hospital day 2
- Patients with IV OR IA Thrombolytic (t-PA) Therapy Administered at This Hospital or Within 24 Hours Prior to Arrival
- Patients with a documented Reason for Not Administering Antithrombotic Therapy by End of Hospital Day 2
Data Elements:
Risk Adjustment: No.
Data Collection Approach: Retrospective data sources for required data elements include administrative data and medical records. Some hospitals may prefer to gather data concurrently by identifying patients in the population of interest. This approach provides opportunities for improvement at the point of care/service. However, complete documentation includes the principal or other ICD-10 diagnosis and procedure codes, which require retrospective data entry.
Data Accuracy: Variation may exist in the assignment of ICD-10 codes; therefore, coding practices may require evaluation to ensure consistency.
Measure Analysis Suggestions: None
Sampling: Yes. Please refer to the measure set specific sampling requirements and for additional information see the Population and Sampling Specifications section.
Data Reported As: Aggregate rate generated from count data reported as a proportion.
Selected References:
- Adams, H., R. Adams, G. Del Zoppo, L. B. Goldstein, Association Stroke Council of the American Heart, and Association American Stroke. "Guidelines for the Early Management of Patients with Ischemic Stroke: 2005 Guidelines Update a Scientific Statement from the Stroke Council of the American Heart Association/American Stroke Association." [In eng]. Stroke 36, no. 4 (Apr 2005): 916-23.
- Adams, H. P., Jr., G. del Zoppo, M. J. Alberts, D. L. Bhatt, L. Brass, A. Furlan, R. L. Grubb, et al. "Guidelines for the Early Management of Adults with Ischemic Stroke: A Guideline from the American Heart Association/American Stroke Association Stroke Council, Clinical Cardiology Council, Cardiovascular Radiology and Intervention Council, and the Atherosclerotic Peripheral Vascular Disease and Quality of Care Outcomes in Research Interdisciplinary Working Groups: The American Academy of Neurology Affirms the Value of This Guideline as an Educational Tool for Neurologists." [In eng]. Stroke 38, no. 5 (May 2007): 1655-711.
- Albers, G. W, P Amarenco, J. D. Easton, R. L. Sacco, and P. Teal. "Antithrombotic and Thrombolytic Therapy for Ischemic Stroke." Chest 119 (2001): 300-20.
- Antithrombotic Trialists, Collaboration. "Collaborative Meta-Analysis of Randomised Trials of Antiplatelet Therapy for Prevention of Death, Myocardial Infarction, and Stroke in High Risk Patients." [In eng]. BMJ 324, no. 7329 (Jan 12 2002): 71-86.
- Brott, T. G., W. M. Clark, S. C. Fagan, J. C. Grotta, L. N. Hopkins, E. C. Jauch, R. E. Latchaw, and S. Starkman. "Stroke: The First Hours. Guidelines for Acute Treatment." National Stroke Association (NSA) (2000).
- Centers for Disease Control and Prevention. "Prevalence and Most Common Causes of Disability among Adults--United States, 2005." [In eng]. MMWR Morb Mortal Wkly Rep 58, no. 16 (May 1 2009): 421-6.
- Chen, Z. M., P. Sandercock, H. C. Pan, C. Counsell, R. Collins, L. S. Liu, J. X. Xie, C. Warlow, and R. Peto. "Indications for Early Aspirin Use in Acute Ischemic Stroke : A Combined Analysis of 40 000 Randomized Patients from the Chinese Acute Stroke Trial and the International Stroke Trial. On Behalf of the Cast and Ist Collaborative Groups." [In eng]. Stroke 31, no. 6 (Jun 2000): 1240-9.
- Coull, B. M., L. S. Williams, L. B. Goldstein, J. F. Meschia, D. Heitzman, S. Chaturvedi, K. C. Johnston, et al. "Anticoagulants and Antiplatelet Agents in Acute Ischemic Stroke: Report of the Joint Stroke Guideline Development Committee of the American Academy of Neurology and the American Stroke Association (a Division of the American Heart Association)." [In eng]. Stroke 33, no. 7 (Jul 2002): 1934-42.
- Eccles, M., N. Freemantle, and J. Mason. "North of England Evidence Based Guideline Development Project: Guideline on the Use of Aspirin as Secondary Prophylaxis for Vascular Disease in Primary Care. North of England Aspirin Guideline Development Group." [In eng]. BMJ 316, no. 7140 (Apr 25 1998): 1303-9.
- "The European Stroke Prevention Study (Esps). Principal End-Points. The Esps Group." [In eng]. Lancet 2, no. 8572 (Dec 12 1987): 1351-4.
- Furie, K. L., S. E. Kasner, R. J. Adams, G. W. Albers, R. L. Bush, S. C. Fagan, J. L. Halperin, et al. "Guidelines for the Prevention of Stroke in Patients with Stroke or Transient Ischemic Attack: A Guideline for Healthcare Professionals from the American Heart Association/American Stroke Association." [In eng]. Stroke 42, no. 1 (Jan 2011): 227-76.
- Gaspoz, J. M., P. G. Coxson, P. A. Goldman, L. W. Williams, K. M. Kuntz, M. G. Hunink, and L. Goldman. "Cost Effectiveness of Aspirin, Clopidogrel, or Both for Secondary Prevention of Coronary Heart Disease." [In eng]. N Engl J Med 346, no. 23 (Jun 6 2002): 1800-6.
- Guyatt, G. H., E. A. Akl, M. Crowther, D. D. Gutterman, H. J. Schuunemann, Therapy American College of Chest Physicians Antithrombotic, and Panel Prevention of Thrombosis. "Executive Summary: Antithrombotic Therapy and Prevention of Thrombosis, 9th Ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines." [In eng]. Chest 141, no. 2 Suppl (Feb 2012): 7S-47S.
- "The International Stroke Trial (Ist): A Randomised Trial of Aspirin, Subcutaneous Heparin, Both, or Neither among 19435 Patients with Acute Ischaemic Stroke. International Stroke Trial Collaborative Group." [In eng]. Lancet 349, no. 9065 (May 31 1997): 1569-81.
- Jauch, E. C., J. L. Saver, H. P. Adams, Jr., A. Bruno, J. J. Connors, B. M. Demaerschalk, P. Khatri, et al. "Guidelines for the Early Management of Patients with Acute Ischemic Stroke: A Guideline for Healthcare Professionals from the American Heart Association/American Stroke Association." [In Eng]. Stroke (Jan 31 2013).
- Johnson, E. S., S. F. Lanes, C. E. Wentworth, 3rd, M. H. Satterfield, B. L. Abebe, and L. W. Dicker. "A Metaregression Analysis of the Dose-Response Effect of Aspirin on Stroke." [In eng]. Arch Intern Med 159, no. 11 (Jun 14 1999): 1248-53.
- Roger, V. L., A. S. Go, D. M. Lloyd-Jones, E. J. Benjamin, J. D. Berry, W. B. Borden, D. M. Bravata, et al. "Heart Disease and Stroke Statistics--2012 Update: A Report from the American Heart Association." [In eng]. Circulation 125, no. 1 (Jan 3 2012): e2-e220.
- Sacco, R. L., R. Adams, G. Albers, M. J. Alberts, O. Benavente, K. Furie, L. B. Goldstein, et al. "Guidelines for Prevention of Stroke in Patients with Ischemic Stroke or Transient Ischemic Attack: A Statement for Healthcare Professionals from the American Heart Association/American Stroke Association Council on Stroke: Co-Sponsored by the Council on Cardiovascular Radiology and Intervention: The American Academy of Neurology Affirms the Value of This Guideline." [In eng]. Stroke 37, no. 2 (Feb 2006): 577-617.
Measure Algorithm: