Release Notes:
Measure Information Form
Version 2016A1
**NQF-ENDORSED VOLUNTARY CONSENSUS STANDARDS FOR HOSPITAL CARE**Measure Information Form
Measure Set: Stroke(STK)
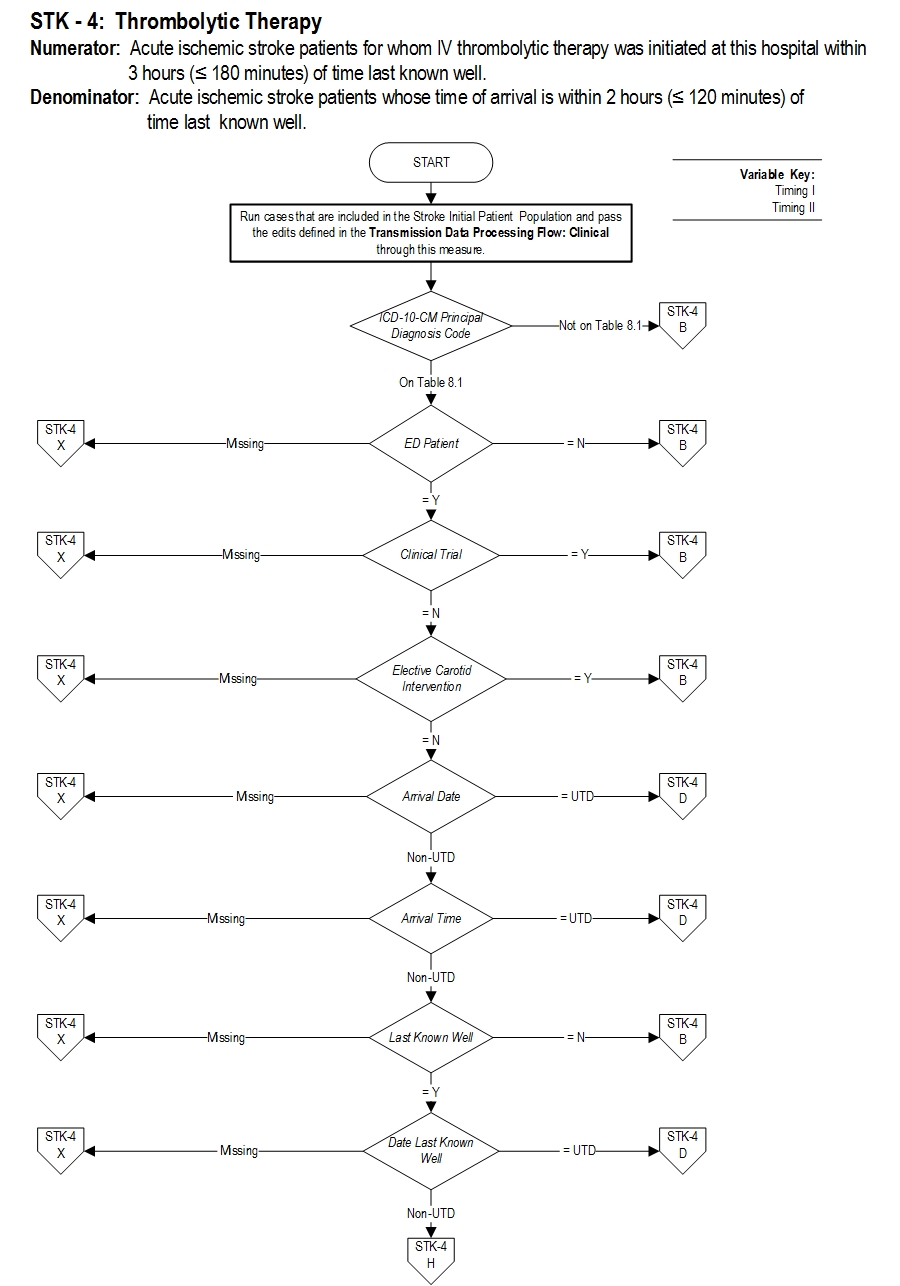
Set Measure ID: STK-4
Performance Measure Name: Thrombolytic Therapy
Description: Acute ischemic stroke patients who arrive at this hospital within 2 hours of time last known well and for whom IV t-PA was initiated at this hospital within 3 hours of time last known well.
Rationale: The administration of thrombolytic agents to carefully screened, eligible patients with acute ischemic stroke has been shown to be beneficial in several clinical trials. These included two positive randomized controlled trials in the United States: The National Institute of Neurological Disorders and Stroke (NINDS) Studies, Part I and Part II. Based on the results of these studies, the Food and Drug Administration (FDA) approved the use of intravenous recombinant tissue plasminogen activator (IV r-TPA or t-PA) for the treatment of acute ischemic stroke when given within 3 hours of stroke symptom onset. A large meta-analysis controlling for factors associated with stroke outcome confirmed the benefit of IV t-PA in patients treated within 3 hours of symptom onset. While controversy still exists among some specialists, the major society practice guidelines developed in the United States all recommend the use of IV t-PA for eligible patients. Physicians with experience and skill in stroke management and the interpretation of CT scans should supervise treatment.
The European Cooperative Acute Stroke Study (ECASS) III trial indicated that intravenous rtPA can be given safely to, and can improve outcomes for, carefully selected patients treated 3 to 4.5 hours after stroke; however, as the NINDS investigators concluded, the earlier that IV thrombolytic therapy is initiated, the better the patient outcome. Therefore, the target for IV t-PA initiation remains within 3 hours of time last known well. The administration of IV thrombolytic therapy beyond 3 hours of stroke symptom onset has not been FDA approved.
Type of Measure: Process
Improvement Noted As: Increase in the rate
Numerator Statement: Acute ischemic stroke patients for whom IV thrombolytic therapy was initiated at this hospital within 3 hours (less than or equal to 180 minutes) of time last known well.
Included Populations: Not applicable
Excluded Populations: None
Data Elements:
Denominator Statement: Acute ischemic stroke patients whose time of arrival is within 2 hours (less than or equal to 120 minutes) of time last known well.
Included Populations:
Discharges with an ICD-10-CM Principal Diagnosis Code for ischemic stroke as defined in Appendix A, Table 8.1
Excluded Populations:
- Patients less than 18 years of age
- Patients who have a Length of Stay greater than 120 days
- Patients enrolled in clinical trials
- Patients admitted for Elective Carotid Intervention
- Time Last Known Well to arrival in the emergency department greater than 2 hours
- Patients with a documented Reason For Extending the Initiation of IV Thrombolytic
- Patients with a documented Reason For Not Initiating IV Thrombolytic
Data Elements:
Risk Adjustment: Yes.
Data Collection Approach: Retrospective data sources for required data elements include administrative data and medical records. Some hospitals may prefer to gather data concurrently by identifying patients in the population of interest. This approach provides opportunities for improvement at the point of care/service. However, complete documentation includes the principal or other ICD-10 diagnosis and procedure codes, which require retrospective data entry.
Data Accuracy: Variation may exist in the assignment of ICD-10 codes; therefore, coding practices may require evaluation to ensure consistency.
Measure Analysis Suggestions: None
Sampling: Yes. Please refer to the measure set specific sampling requirements and for additional information see the Population and Sampling Specifications section.
Data Reported As: Aggregate rate generated from count data reported as a proportion.
Selected References:
- Adams, H., R. Adams, G. Del Zoppo, L. B. Goldstein, Association Stroke Council of the American Heart, and Association American Stroke. "Guidelines for the Early Management of Patients with Ischemic Stroke: 2005 Guidelines Update a Scientific Statement from the Stroke Council of the American Heart Association/American Stroke Association." [In eng]. Stroke 36, no. 4 (Apr 2005): 916-23.
- Adams, H. P., Jr., G. del Zoppo, M. J. Alberts, D. L. Bhatt, L. Brass, A. Furlan, R. L. Grubb, et al. "Guidelines for the Early Management of Adults with Ischemic Stroke: A Guideline from the American Heart Association/American Stroke Association Stroke Council, Clinical Cardiology Council, Cardiovascular Radiology and Intervention Council, and the Atherosclerotic Peripheral Vascular Disease and Quality of Care Outcomes in Research Interdisciplinary Working Groups: The American Academy of Neurology Affirms the Value of This Guideline as an Educational Tool for Neurologists." [In eng]. Stroke 38, no. 5 (May 2007): 1655-711.
- Albers, G. W., P. Amarenco, J. D. Easton, R. L. Sacco, and P. Teal. "Antithrombotic and Thrombolytic Therapy for Ischemic Stroke: The Seventh Accp Conference on Antithrombotic and Thrombolytic Therapy." [In eng]. Chest 126, no. 3 Suppl (Sep 2004): 483S-512S.
- Brott, T. G., W. M. Clark, S. C. Fagan, J. C. Grotta, L. N. Hopkins, E. C. Jauch, R. E. Latchaw, and S. Starkman. "Stroke: The First Hours. Guidelines for Acute Treatment." National Stroke Association (NSA) (2000).
- Centers for Disease Control and Prevention. "Prevalence and Most Common Causes of Disability among Adults--United States, 2005." [In eng]. MMWR Morb Mortal Wkly Rep 58, no. 16 (May 1 2009): 421-6.
- Del Zoppo, G. J., J. L. Saver, E. C. Jauch, H. P. Adams, Jr., and Council American Heart Association Stroke. "Expansion of the Time Window for Treatment of Acute Ischemic Stroke with Intravenous Tissue Plasminogen Activator: A Science Advisory from the American Heart Association/American Stroke Association." [In eng]. Stroke 40, no. 8 (Aug 2009): 2945-8.
- "Diagnosis and Initial Treatment of Ischemic Stroke." Institute for Clinical Systems Improvement (2001).
- Fagan, S. C., L. B. Morgenstern, A. Petitta, R. E. Ward, B. C. Tilley, J. R. Marler, S. R. Levine, et al. "Cost-Effectiveness of Tissue Plasminogen Activator for Acute Ischemic Stroke. Ninds Rt-Pa Stroke Study Group." [In eng]. Neurology 50, no. 4 (Apr 1998): 883-90.
- Guyatt, G. H., E. A. Akl, M. Crowther, D. D. Gutterman, H. J. Schuunemann, Therapy American College of Chest Physicians Antithrombotic, and Panel Prevention of Thrombosis. "Executive Summary: Antithrombotic Therapy and Prevention of Thrombosis, 9th Ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines." [In eng]. Chest 141, no. 2 Suppl (Feb 2012): 7S-47S.
- Hacke, W., G. Donnan, C. Fieschi, M. Kaste, R. von Kummer, J. P. Broderick, T. Brott, et al. "Association of Outcome with Early Stroke Treatment: Pooled Analysis of Atlantis, Ecass, and Ninds Rt-Pa Stroke Trials." [In eng]. Lancet 363, no. 9411 (Mar 6 2004): 768-74.
- Hacke, W., M. Kaste, E. Bluhmki, M. Brozman, A. Davalos, D. Guidetti, V. Larrue, et al. "Thrombolysis with Alteplase 3 to 4.5 Hours after Acute Ischemic Stroke." [In eng]. N Engl J Med 359, no. 13 (Sep 25 2008): 1317-29.
- Hacke, W., M. Kaste, C. Fieschi, D. Toni, E. Lesaffre, R. von Kummer, G. Boysen, et al. "Intravenous Thrombolysis with Recombinant Tissue Plasminogen Activator for Acute Hemispheric Stroke. The European Cooperative Acute Stroke Study (Ecass)." [In eng]. JAMA 274, no. 13 (Oct 4 1995): 1017-25.
- Jauch, E. C., J. L. Saver, H. P. Adams, Jr., A. Bruno, J. J. Connors, B. M. Demaerschalk, P. Khatri, et al. "Guidelines for the Early Management of Patients with Acute Ischemic Stroke: A Guideline for Healthcare Professionals from the American Heart Association/American Stroke Association." [In Eng]. Stroke (Jan 31 2013).
- Kwiatkowski, T. G., R. B. Libman, M. Frankel, B. C. Tilley, L. B. Morgenstern, M. Lu, J. P. Broderick, et al. "Effects of Tissue Plasminogen Activator for Acute Ischemic Stroke at One Year. National Institute of Neurological Disorders and Stroke Recombinant Tissue Plasminogen Activator Stroke Study Group." [In eng]. N Engl J Med 340, no. 23 (Jun 10 1999): 1781-7.
- "Management of Patients with Stroke: Rehabilitation, Prevention and Management of Complications, and Discharge Planning. A National Clinical Guideline.". http://www.sign.ac.uk/guidelines/fulltext/118/.
- Marler, J. R., B. C. Tilley, M. Lu, T. G. Brott, P. C. Lyden, J. C. Grotta, J. P. Broderick, et al. "Early Stroke Treatment Associated with Better Outcome: The Ninds Rt-Pa Stroke Study." [In eng]. Neurology 55, no. 11 (Dec 12 2000): 1649-55.
- Roger, V. L., A. S. Go, D. M. Lloyd-Jones, E. J. Benjamin, J. D. Berry, W. B. Borden, D. M. Bravata, et al. "Heart Disease and Stroke Statistics--2012 Update: A Report from the American Heart Association." [In eng]. Circulation 125, no. 1 (Jan 3 2012): e2-e220.
- Sacco, R. L., R. Adams, G. Albers, M. J. Alberts, O. Benavente, K. Furie, L. B. Goldstein, et al. "Guidelines for Prevention of Stroke in Patients with Ischemic Stroke or Transient Ischemic Attack: A Statement for Healthcare Professionals from the American Heart Association/American Stroke Association Council on Stroke: Co-Sponsored by the Council on Cardiovascular Radiology and Intervention: The American Academy of Neurology Affirms the Value of This Guideline." [In eng]. Stroke 37, no. 2 (Feb 2006): 577-617.
- Saposnik, G., J. Fang, M. K. Kapral, J. V. Tu, M. Mamdani, P. Austin, S. C. Johnston, Network Investigators of the Registry of the Canadian Stroke, and Group Stroke Outcomes Research Canada Working. "The Iscore Predicts Effectiveness of Thrombolytic Therapy for Acute Ischemic Stroke." [In eng]. Stroke 43, no. 5 (May 2012): 1315-22.
- "Tissue Plasminogen Activator for Acute Ischemic Stroke. The National Institute of Neurological Disorders and Stroke Rt-Pa Stroke Study Group." [In eng]. N Engl J Med 333, no. 24 (Dec 14 1995): 1581-7.
- Wardlaw, J. M., V. Murray, E. Berge, and G. J. Del Zoppo. "Thrombolysis for Acute Ischaemic Stroke." [In eng]. Cochrane Database Syst Rev, no. 4 (2009): CD000213.
- U.S. Drug and Food Administration. (2015). “Label- Activase-Food and Drug.”
Measure Algorithm: